Bio Decontamination Market Summary
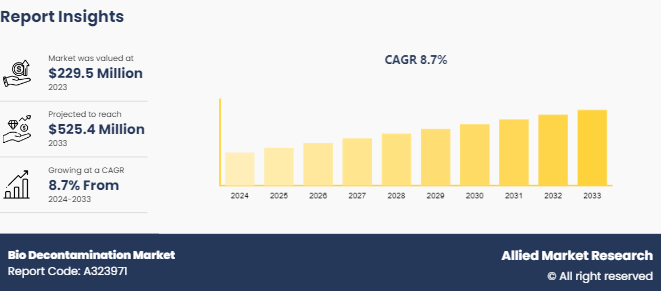
The global bio decontamination market size was valued at $229.5 million in 2023, and is projected to reach $525.4 million by 2033, growing at a CAGR of 8.7% from 2024 to 2033. Increase in the number of hospital-acquired infections (HAIs), growing biopharmaceutical production, and heightened awareness regarding contamination control are driving the demand for bio decontamination solutions across healthcare and life sciences sectors.
What is Meant by Bio decontamination
The major factors driving the growth of the bio decontamination market are increasing awareness about the importance of maintaining sterile environments in healthcare and pharmaceutical sectors is a significant driver, rise in hospital-acquired infections (HAIs) necessitates stringent decontamination protocols and growing pharmaceutical and biotechnology industries demand high standards of cleanliness to ensure product safety and efficacy.
Market Size & Future Outlook
- 2023 Market Size: $229.5 Billion
- 2033 Projected Market Size: $525.45 Billion
- CAGR (2024-2033): 8.64%
- North America: Largest market in 2023
- Asia Pacific: Fastest growing market
Market Introduction and Definition
Bio decontamination refers to the systematic process of eliminating or reducing harmful biological contaminants, including bacteria, viruses, fungi, and spores, from surfaces, equipment, and enclosed environments. This process is achieved through advanced technologies such as vaporized hydrogen peroxide (VHP), chlorine dioxide gas, ultraviolet (UV) light, and other automated room decontamination systems. Bio decontamination solutions are widely used in hospitals, pharmaceutical and biotechnology manufacturing facilities, research laboratories, and cleanrooms to maintain sterile conditions and comply with stringent regulatory standards. These systems help prevent cross-contamination, ensure product safety, protect healthcare workers and patients, and support infection control protocols. As regulatory requirements become more rigorous and the focus on biosafety intensifies, bio decontamination technologies are gaining critical importance in contamination-sensitive industries.
Key Takeaways
- The bio decontamination market share study covers 20 countries. The research includes a segment analysis of each country in terms of value for the projected period.
- More than 1, 500 product literatures, industry releases, annual reports, and other such documents of major bio decontamination industry participants along with authentic industry journals, trade associations' releases, and government websites have been reviewed for generating high-value industry insights.
- The study integrated high-quality data, professional opinions and analysis, and critical independent perspectives. The research approach is intended to provide a balanced view of global markets and assist stakeholders in making educated decisions to achieve their most ambitious growth objectives.
Key Market Dynamics
The bio decontamination market is expanding steadily, driven by increasing emphasis on infection prevention, stringent regulatory requirements, and rising investments in healthcare and life sciences infrastructure. Bio decontamination systems are designed to eliminate harmful microorganisms such as bacteria, viruses, fungi, and spores from controlled environments including hospitals, pharmaceutical manufacturing facilities, biotechnology laboratories, and research centers. Technologies such as hydrogen peroxide vapor (HPV), chlorine dioxide gas, ozone, and ultraviolet (UV) systems are widely adopted to ensure sterility, regulatory compliance, and operational safety.
Drivers
The growing incidence of hospital-acquired infections (HAIs) is a major factor propelling market growth. Healthcare facilities are increasingly adopting automated bio decontamination solutions to minimize cross-contamination and enhance patient safety. Additionally, rapid expansion of pharmaceutical and biotechnology industries, particularly in vaccine and biologics production, has increased the demand for reliable cleanroom sterilization technologies. Stringent regulations regarding contamination control and Good Manufacturing Practices (GMP) further compel organizations to invest in validated and efficient decontamination systems.
Technological advancements also contribute significantly to market expansion. Automated and portable bio decontamination units equipped with real-time monitoring, cycle validation, and remote operation capabilities improve efficiency and reduce manual intervention. Increased awareness regarding biosafety, especially following global health crises, has accelerated the adoption of advanced sterilization technologies across multiple industries.
Opportunities
Integration of automation, IoT-enabled monitoring, and data analytics into decontamination systems presents substantial growth opportunities. Smart systems capable of remote tracking and documentation enhance compliance and operational transparency. Growing investments in research laboratories, expansion of cleanroom facilities in emerging markets, and rising demand for environmentally friendly sterilization agents further create new avenues for manufacturers.
Restraints
High initial investment costs associated with advanced bio decontamination systems may limit adoption, particularly among small and medium-sized facilities. Operational complexities, maintenance requirements, and the need for trained personnel can also pose challenges. Additionally, regulatory approval processes and validation requirements may extend implementation timelines, potentially restraining market growth during the forecast period.
Bio decontamination Market Segmentation
The bio decontamination market is segmented into product, agent, type and end user and region. By product, the market is divided into equipment and consumables. By agent, the market is divided into hydrogen peroxide, chlorine dioxide and nitrogen dioxide. By type, the market is divided into chamber decontamination, and room decontamination. By end user, the market is divided into pharmaceutical and biotechnology companies, and healthcare facilities. Region wise, the market is analyzed across North America, Europe, Asia-Pacific, and LAMEA.
Regional/Country Market Outlook
North America dominated the bio decontamination market share owing to substantial research and development activities, a strong presence of major key players, and well-established biopharmaceutical industry. In addition, according to bio decontamination market opportunity analysis, Asia-Pacific region is expected to register significant growth in the forecast period owing to expanding biopharmaceutical industry, developing healthcare infrastructure and rise in awareness about the hospital acquired infections.
- The Ministry of Health and Family Welfare, India announced funding of $10.93 billion in the Interim Union Budget 2024-25. The funding is allocated to boost the country’s healthcare infrastructure.
- According to 2022 National and State Estimates, by Pharmaceutical Research and Manufacturers of America, in 2021, the U.S. biopharmaceutical industry performs 46% of all biopharmaceuticals basic research, 22% of all biopharmaceuticals applied research, and 15% of all biopharmaceutical development.
- According to the UK Government, the turnover of the UK life sciences industry registered growth of 13% from the financial year 2020-2021 to the financial year 2021-2022.
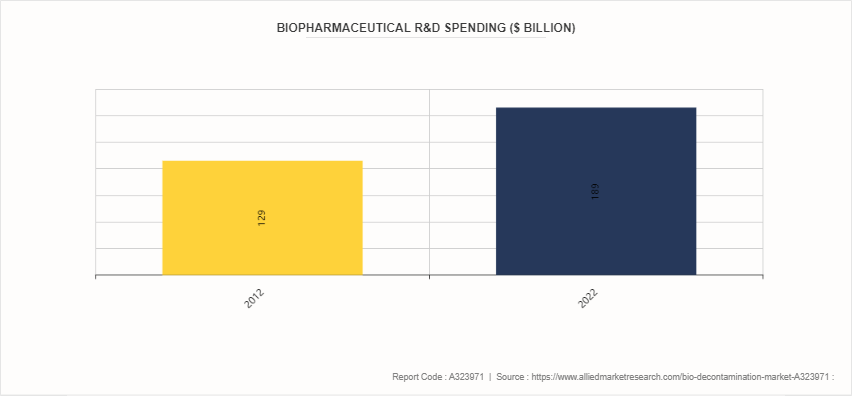
Rise in Research and Development Investment in Biopharmaceutical Industry
The rise in biopharmaceutical R&D spending is poised to significantly impact the bio decontamination market in several ways. Increased investment in research and development by biopharmaceutical companies leads to the establishment of more laboratories and production facilities, all of which require stringent contamination control measures to ensure the integrity and safety of their products. This heightened focus on cleanliness and sterile environments will drive the demand for advanced bio decontamination solutions, such as vaporized hydrogen peroxide systems, UV-C light sterilization, and other innovative technologies. Furthermore, as biopharmaceutical companies push the boundaries of scientific discovery, they encounter new and complex contamination challenges, necessitating more sophisticated and effective decontamination strategies
Industry Trends
- According to a 2024 article by the Center of Disease Control and Prevention, everyday about 1 in 31 hospital patients has at least one healthcare-associated infection. The rise in healthcare-associated infection is expected to drive the demand for effective bio decontamination solutions.
- According to a 2022 article by the European Federation of Pharmaceutical Industries and Association, in 2021, the European pharmaceutical industry invested around $49.082 billion on research and development activities. Higher R&D investment often leads to an increase in the number of clinical trials. This surge in clinical trials requires stringent bio-decontamination protocols to ensure the safety and sterility of laboratory environments, equipment, and facilities.
Key End-User Segments Fueling the Bio Decontamination Market
The bio decontamination market is driven by several primary end users across healthcare, life sciences, and industrial sectors. Hospitals and healthcare facilities represent one of the largest end-user segments, as infection prevention remains a top priority. These facilities utilize bio decontamination systems to sterilize operating rooms, intensive care units, isolation wards, and patient rooms. Automated hydrogen peroxide vapor (HPV) and ultraviolet (UV) systems are widely adopted to reduce hospital-acquired infections (HAIs) and ensure compliance with stringent hygiene standards. The increasing focus on patient safety and regulatory adherence continues to accelerate technology adoption in healthcare environments.
Pharmaceutical and biotechnology companies form another critical segment contributing significantly to market growth. These organizations rely on advanced bio decontamination solutions to maintain sterile cleanrooms, production suites, isolators, and research laboratories. Strict regulatory frameworks governing Good Manufacturing Practices (GMP) require validated decontamination processes to prevent contamination during drug development and manufacturing. The rapid growth of biologics, vaccine production, and cell and gene therapy research further strengthens demand for high-efficiency and automated systems.
Research laboratories and academic institutions also play a vital role in market expansion. Universities, government research centers, and biosafety laboratories implement bio decontamination technologies to ensure safe handling of hazardous biological agents. Increased funding for life sciences research and public health preparedness initiatives supports consistent investment in contamination control systems.
In addition, food processing facilities and contract research organizations (CROs) are emerging as important end users. These entities adopt bio decontamination technologies to maintain product integrity, comply with safety regulations, and reduce contamination risks. The broadening application scope across diverse industries continues to fuel sustained growth in the bio decontamination market.
Patent Analysis for the Bio Decontamination Market
According to recent global patent filing trends between 2022 and 2024, innovation in the bio decontamination market remains concentrated in technologically advanced regions. China accounts for a significant share of bio decontamination-related patents, reflecting its expanding capabilities in sterilization equipment manufacturing, automation technologies, and cleanroom solutions. The United States follows closely, driven by strong research activity in healthcare, biotechnology, and pharmaceutical contamination control systems. Filings through the Patent Cooperation Treaty (PCT) system also represent a notable portion, highlighting the intent of companies to secure multi-regional intellectual property protection.
The European Patent Office and Japan contribute steadily to global filings, particularly in hydrogen peroxide vapor (HPV) systems, ultraviolet (UV-C) disinfection technologies, and automated room decontamination units. Other active contributors include South Korea and Germany, both recognized for advancements in precision engineering and industrial sterilization systems. Patent activity is largely focused on innovations in automated cycle validation, real-time environmental monitoring, eco-friendly sterilants, and integration of IoT-enabled smart control systems. This distribution of patents reflects a competitive landscape where companies prioritize efficiency, safety, regulatory compliance, and sustainability.
Impact of Evolving Consumer and Industry Preferences on Bio Decontamination Market Trends
Evolving preferences among healthcare providers, pharmaceutical manufacturers, and research institutions are significantly influencing market trends. One of the most prominent shifts is the increasing demand for automated and touchless decontamination solutions. End users prioritize systems that minimize human intervention, reduce contamination risks, and ensure reproducible sterilization outcomes. This has accelerated the adoption of fully automated hydrogen peroxide vapor systems and mobile UV disinfection units.
There is also growing emphasis on faster cycle times and operational efficiency. Facilities seek solutions that can achieve high-level disinfection with minimal downtime, particularly in hospitals and high-throughput pharmaceutical environments. Compact, portable systems are gaining popularity due to their flexibility and ease of deployment across multiple rooms or facilities.
Sustainability considerations are further reshaping bio decontamination market demand. Organizations increasingly prefer environmentally friendly sterilization agents that leave minimal residue and reduce chemical exposure risks. Additionally, digital integration plays a critical role in decision-making. Users favor systems equipped with cloud-based monitoring, automated reporting, and compliance documentation features to meet stringent regulatory standards.
Collectively, these evolving preferences are redefining product development strategies and accelerating innovation across the global bio decontamination market.
Competitive Landscape
The major players operating in the bio decontamination market include Fedegari Autoclavi SpA, Steris PLC, TOMI Environmental Solutions, Inc, Ecolab Inc, JCE Biotechnology, DIOP GmbH & Co. KG, Howorth Air Technology Ltd., Zhejiang TAILIN Bioengineering Co., LTD, AM Instruments srl, Solidfog Technologies. Other players in the bio decontamination market include Noxilizer, Inc., ClorDiSys Solutions, Inc., Amira SRL, Controlled Contamination Services, and Tecomak.
Leading Companies and Strategic Initiatives Shaping the Bio Decontamination Market
Key players operating in the bio decontamination market include Fedegari Autoclavi SpA, STERIS plc, TOMI Environmental Solutions, Inc., Ecolab Inc., JCE Biotechnology, DIOP GmbH & Co. KG, Howorth Air Technology Ltd., Zhejiang TAILIN Bioengineering Co., LTD, AM Instruments srl, and Solidfog Technologies. Other notable participants include Noxilizer, Inc., ClorDiSys Solutions, Inc., Amira SRL, Controlled Contamination Services, and Tecomak. These companies maintain strong market positions through comprehensive product portfolios, global distribution networks, and continuous technological innovation.
A central strategy among leading firms is the development of advanced, automated bio decontamination systems. Companies such as STERIS plc and Fedegari Autoclavi SpA focus on integrated sterilization and cleanroom solutions that comply with stringent regulatory standards in pharmaceutical and healthcare industries. TOMI Environmental Solutions emphasizes hydrogen peroxide-based disinfection technologies, while Noxilizer, Inc. specializes in nitrogen dioxide sterilization systems designed for rapid, low-temperature applications.
Strategic partnerships, acquisitions, and geographic expansion are also widely adopted to strengthen competitive positioning. Ecolab Inc. leverages its global presence in water, hygiene, and infection prevention solutions to expand its bio decontamination offerings. European players such as Howorth Air Technology Ltd. and DIOP GmbH & Co. KG focus on cleanroom engineering and turnkey contamination control solutions. Meanwhile, emerging Asian manufacturers, including Zhejiang TAILIN Bioengineering Co., LTD, are increasing investments in research and development to enhance automation and cost efficiency. Collectively, these strategies reinforce innovation, regulatory compliance, and sustained growth across the global bio decontamination market.
What are the Recent Development in the Bio Decontamination Industry
- In July 2022, Ecolab Inc announced that HPV-AQ 35% hydrogen peroxide disinfectant received an approval from the European Chemicals Agency (ECHA) Biocidal Products Committee (BPC) for a BPR EU (Union) authorization. Ecolab’s Bioquell HPV-AQ is authorized for use in all territories of the European Union and the European Economic Area.
- In January 2024, Steris plc introduced a new line of hydrogen peroxide-based bio decontamination systems with improved efficiency and faster cycle times, addressing rising demand for quicker and more effective sterilization solutions in healthcare and biotech settings.
Key Sources Referred
- National Library of Medicine
- Center of Disease Control and Prevention
- Pharmaceutical Research and Manufacturers of America
- Government of UK
- Ministry of Health and Family Welfare, India
- European Federation of Pharmaceutical Industries and Association
What are the Key Benefits for Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the bio decontamination market analysis from 2024 to 2033 to identify the prevailing bio decontamination market opportunities.
- The bio decontamination market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the bio decontamination market segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global bio decontamination market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the bio decontamination market players.
- The report includes the analysis of the regional as well as global bio decontamination market trends, key players, market segments, application areas, and market growth strategies.
Bio Decontamination Market Report Highlights
| Aspects | Details |
| Market Size By 2033 | USD 525.4 Million |
| Growth Rate | CAGR of 8.7% |
| Forecast period | 2024 - 2033 |
| Report Pages | 280 |
| By Product |
|
| By Agent |
|
| By Type |
|
| By End User |
|
| By Region |
|
| Key Market Players | TOMI Environmental Solutions Inc, Howorth Air Technology Ltd, Fedegari Autoclavi SpA, Ecolab Inc, DIOP GmbH & Co KG, AM Instruments SRL, JCE Biotechnology, Solidfog Technologies, Zhejiang TAILIN Bioengineering Co., LTD, STERIS plc |
The global bio decontamination market size was valued at $229.5 million in 2023.
The market value of Bio Decontamination Market is projected to reach $525.4 million by 2033
The forecast period for Bio Decontamination Market is 2024-2033.
The base year is 2023 in Bio Decontamination Market
Major key players that operate in the Bio Decontamination Market are Fedegari Autoclavi SpA, Steris PLC, TOMI Environmental Solutions, Inc, and Ecolab Inc,
Loading Table Of Content...



