Capecitabine Market Research, 2033
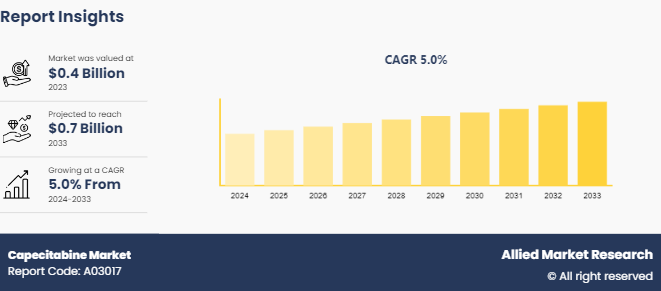
The global capecitabine market size was valued at $0.4 billion in 2023, and is projected to reach $0.7 billion by 2033, growing at a CAGR of 5% from 2024 to 2033. The major factors driving the growth of the capecitabine market are surge in prevalence of cancer, improved healthcare access, rise in research and development to enhancing the efficacy and safety profiles of capecitabine.
Capecitabine is an oral chemotherapy medication used to treat different types of cancer. It functions as a prodrug, which means it is converted into its active form inside the body. It is designed to generate 5-fluorouracil (5-FU) within tumor tissues. It allows for targeted anticancer activity while reducing exposure to healthy tissues. Capecitabine was developed to provide convenient alternative to intravenous 5-FU therapy. The drug is absorbed through the gastrointestinal tract and undergoes a three-step enzymatic conversion process in the liver and tumor tissues. The final step converts it into 5-FU, which interferes with DNA synthesis in rapidly dividing cancer cells. This disruption prevents tumor growth and can lead to cancer cell death.
Market Introduction and Definition
Capecitabine is an oral chemotherapy medication used in the treatment of various cancers, including breast, colon, and rectal cancers. It belongs to a class of drugs known as antimetabolites, which work by interfering with the growth and spread of cancer cells. Capecitabine is a prodrug, meaning it is converted into its active form, 5-fluorouracil (5-FU), in the body. Once converted, 5-FU inhibits the synthesis of DNA and RNA in cancer cells, ultimately leading to their death. Unlike traditional chemotherapy drugs administered intravenously, capecitabine is taken orally, usually in the form of tablets. This offers convenience to patients and allows for outpatient treatment in many cases. Furthermore, capecitabine has shown promise in combination therapies, enhancing its effectiveness in targeting cancer cells while minimizing adverse effects. Its versatility in combination with other drugs or therapies allows for tailored treatment approaches based on individual patient needs and cancer characteristics. For instance, oxaliplatin and capecitabine is a chemotherapy combination known as Xelox, which is used for the treatment of bowel cancer (colorectal cancer) , cancer of the food pipe (oesophagus) , and stomach cancer.
The medication is commonly prescribed for metastatic colorectal cancer, either as monotherapy or in combination with other chemotherapy agents. It is also used in the treatment of metastatic breast cancer, particularly in patients who have not responded to other chemotherapy regimens such as anthracyclines or taxanes. In some cases, capecitabine is used as adjuvant therapy following surgery to reduce the risk of cancer recurrence.
One of the key advantages of capecitabine is its oral administration. Patients can take the medication at home, which reduces the need for hospital visits and intravenous infusions. This improves patient convenience and quality of life, especially for individuals who need long-term chemotherapy. However, adherence to dosing schedules is critical to ensure treatment effectiveness.
Capecitabine is associated with side effects. Common adverse effects include hand-foot syndrome (redness, swelling, and pain in the palms and soles), diarrhea, nausea, fatigue, and mouth sores. The serious ones include bone marrow suppression, which can lead to reduced white blood cell counts and increased infection risk. Dose adjustments are often necessary based on patient tolerance and kidney function. Capecitabine dosing is calculated based on body surface area and is administered in cycles, usually taken twice daily for two weeks followed by a one-week rest period. Close monitoring by healthcare providers is essential to manage side effects and adjust treatment as needed.
Capecitabine has become an important part of modern oncology protocols. It is available in both branded and generic forms, increasing accessibility in many countries. The development of generic versions has significantly reduced treatment costs, especially in low- and middle-income regions. Its targeted mechanism of action, convenience of administration, and established effectiveness in colorectal and breast cancers make it a widely used and valuable anticancer agent. Despite potential side effects, proper monitoring and dose management allow many patients to benefit from its therapeutic potential.
Key Takeaways
- The capecitabine market study covers 20 countries. The research includes a segment analysis of each country in terms of value ($million) for the projected period 2023-2032.
- More than 1, 500 product literatures, industry releases, annual reports, and other such documents of major energy storage system industry participants along with authentic industry journals, trade associations' releases, and government websites have been reviewed for generating high-value industry insights.
- The study integrates high-quality data, professional opinions and analysis, and critical independent perspectives. The research approach is intended to provide a balanced view of global markets and to assist stakeholders in making educated decisions to achieve their most ambitious growth objectives.
Key Market Dynamics
Drivers
Rise in global prevalence of colorectal and breast cancers and increase in cancer incidence due to aging populations, lifestyle factors, and improved diagnostic capabilities expands the patient base that requires chemotherapy treatment drive the growth of the capecitabine market. Higher diagnosis rates directly contribute to the market growth as capecitabine is a standard therapy option for the treatment of different types of cancers.
Further, preference for oral chemotherapy over intravenous treatment also fuels the market growth. Oral drugs like capecitabine provide convenience, reduce hospital visits, and lower infusion-related costs. This aligns with broader healthcare trends favoring outpatient care and patient-centered treatment approaches. The availability of generic formulations is also a significant factor driving the growth of the market. Governments and healthcare providers often prefer generics to reduce treatment expenditure.
Moreover, ongoing research and combination therapy protocols support continuous use of capecitabine. It is frequently combined with other chemotherapy agents or targeted therapies to enhance treatment outcomes, sustaining its relevance in oncology guidelines.
Restraints
Risk of adverse side effects limit the use of capecitabine market. Further, increase in competition from newer targeted therapies and immunotherapies and advancements in precision oncology have introduced alternative treatments that may offer improved efficacy or reduced side effects compared to traditional chemotherapy agents like capecitabine. Regulatory challenges and pricing pressures also influence market dynamics. In many countries, strict drug approval processes and pricing controls limit profit margins for pharmaceutical manufacturers. Furthermore, the widespread availability of generics intensifies price competition. This deters the growth of the market.
Opportunities
Expansion of healthcare infrastructure and improvements in cancer screening programs in Asia-Pacific, Latin America, and parts of Africa create new patient pools. Increase in government spending on oncology care can further support demand. Genetic testing to identify patients who are more likely to respond to fluoropyrimidine-based therapies may optimize treatment outcomes and encourage continued use of capecitabine. Research into novel drug delivery systems and supportive care strategies also presents potential for improved tolerability. Enhanced formulations that reduce side effects could strengthen patient adherence and clinical acceptance.
How Is Artificial Intelligence Transforming the Capecitabine Market?
Artificial intelligence plays a major role in transforming the market for capecitabine, particularly in oncology research, patient selection, and treatment optimization. It is used in drug development and clinical research. AI-powered platforms analyze datasets from clinical trials, electronic health records, and genomic studies to identify patterns in patient response. This helps researchers determine which patient groups benefit most from capecitabine-based therapies. AI tools can also assist in designing more efficient clinical trials by identifying eligible patients faster and predicting trial outcomes with greater accuracy.
AI is also transforming personalized medicine. Capecitabine is converted into 5-fluorouracil (5-FU) inside the body, and patient response can vary depending on genetic factors such as enzyme activity. AI algorithms can analyze genetic profiles, tumor characteristics, and prior treatment data to predict toxicity risks or treatment effectiveness. This allows oncologists to adjust dosages and select combination therapies more precisely, reducing adverse effects while maintaining efficacy.
In addition, AI-driven imaging analysis helps improve cancer diagnosis and monitoring. Machine learning systems can evaluate radiology images to measure tumor size and progression accurately. These insights help clinicians assess the response of the patient to capecitabine treatment and determine whether therapy should continue, be modified, or be combined with other agents.
Patient Population
Colorectal cancer has seen an increase in incidence rates in recent years. One significant factor contributing to this growth is the aging population, as older individuals are more susceptible to developing cancer due to factors such as accumulated genetic mutations and prolonged exposure to environmental carcinogens. In addition, lifestyle factors such as diets high in red and processed meats, low fiber intake, sedentary lifestyles, obesity, and smoking have been linked to an increased risk of colorectal cancer. As these unhealthy lifestyle choices become more prevalent globally, particularly in urbanized and industrialized regions, the incidence of colorectal cancer continues to rise. Moreover, improvements in screening and diagnostic technologies have led to earlier detection of colorectal cancer cases, which, while beneficial for patients, also contribute to the growing patient population as more cases are identified.
What are the Major Factors Driving the Growth of the Capecitabine Market?
Rise in prevalence of cancer, surge in geriatric population, and high research and development activities to evaluate new pharmacological application of capecitabine are the major factors driving the growth of the market. There is surge in demand for the effective treatment option with growing number of cancer patients globally. For instance, according to the National Cancer Institute, it was estimated that in 2024, 2, 001, 140 new cases of cancer will be diagnosed in the U.S. Capecitabine, being an oral chemotherapy medication with demonstrated efficacy against various types of cancers, including breast, colon, and rectal cancers, has become a key player in the oncology landscape. Its convenience of oral administration provides a significant advantage, particularly in settings where intravenous chemotherapy may be less feasible or accessible. Thus, increase in research and development activities on capecitabine and rise in prevalence of cancer is expected to drive the capecitabine market size.
Market Segmentation
The global capecitabine market is segmented into indication, drug type, end user, and region. By indication, the market is divided into colorectal cancer, breast cancer, and others. By drug type, market is divided into branded and generic. By end user, the market is divided into hospitals, homecare, specialty centers, and others. Region wise, the market is analyzed across North America, Europe, Asia-Pacific, and LAMEA.
Regional/Country Market Outlook
In developed regions such as North America dominated capecitabine market share owing to robust healthcare systems, well-established pharmaceutical industries, and high levels of cancer awareness and screening. These regions typically account for a significant share of global capecitabine market, driven by the widespread adoption of the drug in cancer treatment protocols and the presence of favorable reimbursement policies that facilitate patient access to the medication. According to capecitabine market opportunity analysis emerging markets, particularly in Asia-Pacific and Latin America, the capecitabine market is experiencing rapid growth due to increasing cancer incidence rates, expanding access to healthcare services, and rising healthcare expenditure. Governments and healthcare providers in these regions are increasingly recognizing the importance of oral chemotherapy drugs such as capecitabine in improving treatment outcomes and patient convenience, leading to greater adoption of the medication in clinical practice.
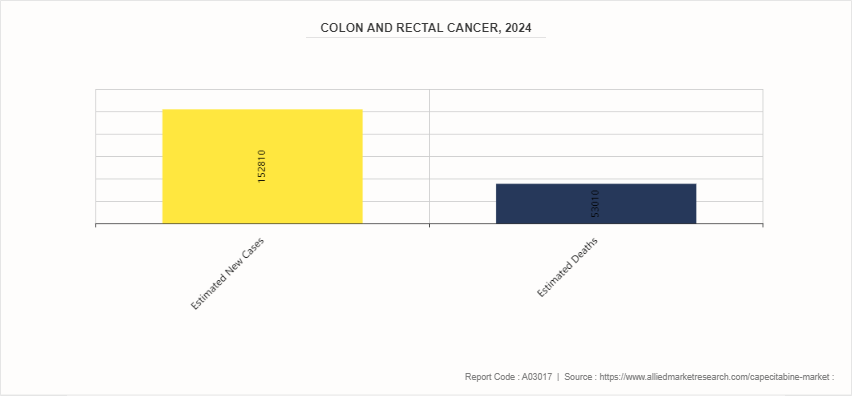
- According to the American Cancer Society estimates that 152, 810 people in the U.S. will be diagnosed with colorectal cancer in 2024.
- According to 2024 article by the National Library of Medicine, it was reported that in 2020 breast cancer accounted for 13.5% of all new cancer cases in India.
- As per 2024 article by World Health Organization, it was reported that breast cancer is the most common cancer prevalent women with an estimated incidence of 604, 900 in 2022.
- The Government of India stated that constant increase in public health expenditure year-on-year. In 2022, expenditure was $63 billion, or 2.1% of GDP, putting the government on track to reach its target of 2.5% by 2025.
Industry Trends
- According to the Center of Disease Control and Prevention, it was estimated that 152, 810 new cases of colon and rectal cancer are expected to be diagnosed in 2024.
- According to 2024 report by the National Cancer Institute, it was reported that the combination of capecitabine and oxaliplatin (CAPOX) is an accepted standard therapy in patients with metastatic colorectal cancer.
- According to 2023 article by the National Library of Medicine, a randomized phase III study was conducted to evaluate capecitabine in combination with either lapatinib or pyrotinib in patients with HER2-positive breast cancer or metastatic breast cancer who previously received trastuzumab, taxanes, and/or anthracyclines. The study concluded that the combination therapy of capecitabine with pyrotinib resulted in the higher survival rate as compared to combination of capecitabine with lapatinib.
- According to 2021 article by the National Library of Medicine, it was reported that capecitabine is widely used in either monotherapy or in combination therapy for treating colorectal cancer.
Competitive Landscape
The major players operating in the capecitabine market include F. Hoffmann-La Roche Ltd., Mylan N.V, Teva Pharmaceutical Industries Ltd., Sanofi, Pfizer Inc., Dr. Reddy’s Laboratories Ltd., Novartis AG, Bayer AG, Eli Lilly and Company, and Merck & Co., Inc. Other players in capecitabine market include GlaxoSmithKline Pharmaceuticals, Sun Pharmaceutical Industries Ltd, Aurobindo Pharma, Lupin, and Alkem Labs Ltd.
Recent Key Strategies and Developments
- In February 2021, Dr. Reddy’s Laboratories Ltd. announced the launch of capecitabine tablets, USP a therapeutic equivalent generic version of Xeloda (capecitabine) tablets approved by the U.S. Food and Drug Administration (USFDA) .
- In December 2022, Shilpa Medicare Ltd. announced the launch of the Capecitabine 1000 MG dispersible tablet with novel technology of faster dispersion within 90 seconds. The product is backed by required scientific proof and comparative bioequivalence studies and is approved by Central Drugs Standard Control Organisation (CDSCO) —the National Regulatory Authority (NRA) of India, responsible for ensuring the safety, efficacy, and quality of medical products in the country. The product is used for treatment of colorectal and metastatic breast cancer.
Key Sources Referred
- World Health Organization
- Ministry of Health, India
- Center of Disease Control and Prevention
- National Library of Medicine
- International Trade Administration
- Europa Donna
- U.S. Department of Commerce
- American Cancer Society
- American Association for Cancer Research
- National Cancer Institute
Key Benefits For Stakeholders
- This report provides a quantitative analysis of the capecitabine market share, current trends, estimations, and dynamics of the capecitabine market analysis from 2024 to 2033 to identify the prevailing capecitabine market opportunities.
- The market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the capecitabine market segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global capecitabine market trends, key players, market segments, application areas, and market growth strategies.
Capecitabine Market Report Highlights
| Aspects | Details |
| Market Size By 2033 | USD 0.7 Billion |
| Growth Rate | CAGR of 5% |
| Forecast period | 2024 - 2033 |
| Report Pages | 280 |
| By Indication |
|
| By Drug Type |
|
| By End User |
|
| By Region |
|
| Key Market Players | Mylan N.V., Merck & Co., Inc., Pfizer Inc., F. Hoffmann-La Roche Ltd., Dr. Reddy’s Laboratories Ltd., Sanofi, Teva Pharmaceutical Industries Ltd., Eli Lilly and Company., Bayer AG, Novartis AG |
Analyst Review
Capecitabine, a chemotherapy drug used primarily in the treatment of colorectal and breast cancers, is witnessing significant growth due to the rising incidence of these cancers globally. In addition, increasing awareness of cancer treatment options, advancements in oncological therapies and ongoing research and development efforts aimed at enhancing the efficacy and safety profiles of capecitabine also significantly contribute in the growth of the market. However, challenges such as stringent regulatory requirements and potential side effects associated with the capecitabine may hinder market growth.
The total market value of Capecitabine Market is $0.4 billion in 2023.
The base year is 2023 in Capecitabine Market
The forecast period for Capecitabine Market is 2024-2033.
The market value of Capecitabine Market is projected to reach $0.7 billion by 2033
Major key players that operate in the Capecitabine Market are F. Hoffmann-La Roche Ltd., Mylan N.V, Teva Pharmaceutical Industries Ltd., and Sanofi.
Loading Table Of Content...



