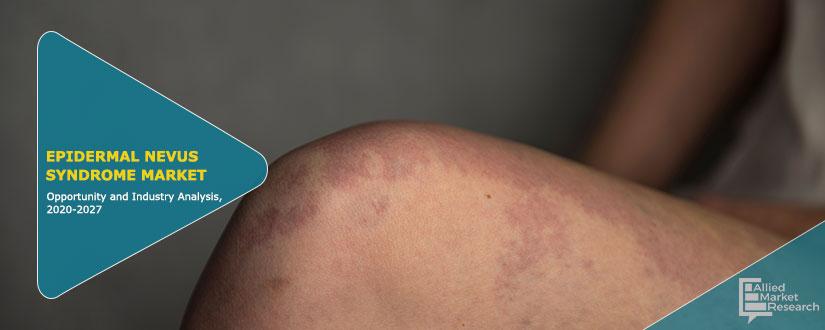
Epidermal nevus syndromes (ENSs) are a group of rare complex disorders characterized by the presence of skin lesions known as epidermal nevi associated with additional extra-cutaneous abnormalities, most often affecting the brain, eye and skeletal systems. Epidermal nevi are overgrowths of structures and tissue of the epidermis, the outermost layer of the skin. Different types of epidermal nevi can vary in size, number, location, distribution, and appearance. Neurological abnormalities that can be associated with ENSs can include seizures, cognitive impairment, developmental delays, and paralysis of one side of the body (hemiparesis). Skeletal abnormalities can include abnormal curvature of the spine, malformation of the hip and abnormalities of the arms and legs (e.g., underdevelopment or absence or overgrowth of limbs).
COVID-19 scenarioanalysis:
Pharmaceutical and biotechnological companies together with governments around the globe are working to combat the COVID-19 outbreak, from supporting the development of vaccines to scheduling medicines supply chain challenges. Around 115 vaccine candidates and other 155 molecules are in the R&D pipeline. Furthermore, the commonly used drugs, such as hydroxychloroquine, have observed huge boost in demand for the management of COVID-19. In addition, increase has been experienced in the demand for COVID-19 management drugs, which is expected to offer potential opportunity for manufacturers of drugs, as many developed countries are short of these drugs. Owing to the growing demand for vaccine and treatment drugs for COVID-19, the pharmaceutical and biotechnology industry is anticipated to witness a substantial growth in the next few years.
Top impacting factors:market scenario analysis, trends, drivers, and impact analysis
The major factor affecting the market include increase in research and development activities to introduce treatment and diagnostic methods to gain higher revenue during the forecast period. This allows the companies to create cost-effective methods and treatment options.Surge in the number of product launches, which meetthe demand posed by the market is anticipated to boost the market in the coming years.Increase in the prevalence of epidermal nevus syndrome associated cases would help the market propel in the next few years. However, lesser medical trained staffs may hamper the growth of the market.
Increase in patient population
Rise in patient population would help the market flourish during the forecast period. For instance, in a recent review published by Rogers and associates, of 131 patients with epidermal nevi and epidermal nevus syndrome, one-third of the patients had the nevus sebaceous type, 60% had the noninflammatory type, 6% had inflammatory linear verrucous epidermal nevus, and only 2 had nevus comedonicus.Around 35 patients with epidermal nevus syndrome seen at the National Institute of Pediatrics in Mexico City during a 31-year period are described. This syndrome represented 7.9% of 443 patients with epidermal nevi; its relative frequency was 1 case per 11,928 pediatric patients and 1 case per 1080 dermatologic patients. Nine epidermal nevus syndrome patients (26%) had Proteus syndrome. Sebaceous nevus syndrome was found in six patients (17%), while the nevus comedonicus syndrome was found in three (8%).
Surge in number of mergers and acquisitions
Increase in the number of mergers and acquisitions is anticipated toboost the marketgrowth in the coming years. For instance, in November 2020, LEO Pharma signed a research collaboration deal with PellePharm that commits up to $760 million for late-stage trials and includes an option for LEO to acquire the small rare disease company outright at some point in the future.
The deal began with $70 million in equity financing and R&D support to fund a global phase 3 trial for PellePharm’s patidegib, a hedgehog pathway-inhibiting topical gel for the prevention of Gorlin syndrome, a rare genetic disease that makes people more prone to develop basal-cell carcinomas.In return for a minority stake in the company, PellePharm could receive up to $690 million upon completion of certain regulatory and commercial milestones, in addition to double-digit royalty payments.
Key benefits of the report:
- This study presents the analytical depiction of the global epidermal nevus syndrome industry along with the current trends and future estimations to determine the imminent investment pockets.
- The report presents information related to key drivers, restraints, and opportunities along with detailed analysis of the global epidermal nevus syndrome market share.
- The current market is quantitatively analyzed to highlight the global epidermal nevus syndrome market growth scenario.
- Porter’s five forces analysis illustrates the potency of buyers & suppliers in the epidermal nevus syndromemarket.
- The report provides a detailed global epidermal nevus syndrome market analysis based on competitive intensity and how the competition will take shape in coming years.
Questions answered in the epidermal nevus syndrome market research report:
- Which are the leading players active in the epidermal nevus syndrome market?
- What are the current trends that will influence the market in the next few years?
- What are the driving factors, restraints, and opportunities of the market?
- What are the projections for the future that would help in tasking further strategic steps?
Epidermal Nevus Syndrome Market Report Highlights
| Aspects | Details |
| By Treatment |
|
| By End User |
|
| By Distribution Channel |
|
| By Region |
|
| Key Market Players | Lynton Lasers Ltd., Fosun Pharmaceutical (Group) Co.,, Siemens Healthineers AG,, Syneron Medical Ltd.,, LUTRONIC,, Solta Medical,, Hologic Inc.,, STRATA Skin Sciences,, GE Healthcare,, Koninklijke Philips N.V., |
Loading Table Of Content...