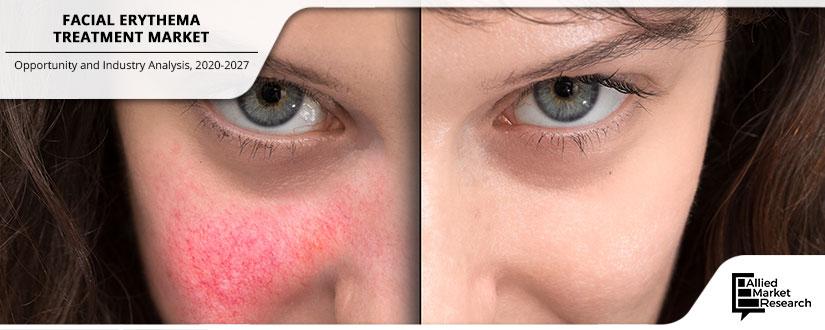
Facial erythema is a condition which causes facial redness in fair-skinned individuals due to the occurrence of cutaneous blood vessel dilation and increased blood flow to the skin facial erythema caused by the various factor such as the common skin diseases including perioral dermatitis, rosacea, and systemic disorder. Facial erythema is causes due to skin injury and this condition mostly seen in fair-skinned people. There are various types of facial erythema occurred in the skin such as rosacea and dermatitis are few inflammatory skin disease. Rosacea is a most common skin disease, which causes redness and visible blood vessels on the face. But there is no cure, treatments can control and reduce the signs and symptoms related with the disease. With the increasing burden of disease and more common occurrence in changing lifestyle, increasing the awareness among the people and physicians have also increased. There are various types of medicines available in the facial erythema treatment market such as antihistamines, antibiotics, calcineurin inhibitors, emollients, antifungal corticosteroids, etc., out of which antibiotics such as metronidazole is most commonly used which can help to prevent the number of acne pimples and bacterial inflammation. The high demand for antibiotics in the treatment of rosacea is the main factor for the growth of the market.
COVID-19 Scenario Analysis:
Pharmaceutical and biotech companies together with governments around the globe are working to address the COVID-19 outbreak, from supporting the development of vaccines to planning for medicines supply chain challenges. Currently, around 115 vaccine candidates and 155 molecules are in the R&D pipeline. Moreover, commonly used drugs such as Hydroxychloroquine have witnessed dramatic surge in demand for the management of COVID-19. Such high demand for these drugs has presented huge opportunity for manufacturers of COVID-19 management drugs, as many developed countries are short of these drugs. Owing to the demand for vaccine and treatment drugs for COVID-19, the pharmaceutical and biotechnology industry is expected to witness a significant growth in the future.
The ongoing outbreak of COVID-19 impacted facial erythema treatment market. The skin and coronavirus interactions, as well as the consequences to the skin and mucous membranes of increased personal hygiene measures, should be recognized by dermatologists and their co-workers. The use of preventive measures, such as emollients, barrier creams, and moisturizers, is important in avoiding skin complications aggravated by preventive measures during the pandemic.
Top impacting factors: Market Scenario Analysis, Trends, Drivers and Impact Analysis:
North America dominates the facial erythema treatment market due to the rising adoption of the treatments, the rising number of fair population triggers the growth of the market in this region. Europe is the second largest market share due to the growing consumption of the alcohol and increasing research and development activities. Whereas, Asia-Pacific is expected to contribute the largest market share due to surging R&D activities, changing lifestyle, rising growth of the population, increasing the number of targeted patients.
In 2018, “National Rosacea Society”, reported that, 737,960,000 people affected with rosacea while the 420,280,000 people was diagnosed from rosacea. Also it reported that approximately 16 million Americans suffer from rosacea. Such trends are pushing more patients suffering from facial erythema to approach clinics and hospitals,
The major factors that drive the growth of facial erythema treatment market such as growing rate of patient population, rising alcohol consumption, rising the launch of new drug products, technological developments and increasing smoking population. The factor such as increasing development in generic drug product and stringent government regulation that are implemented on the approvals of the product that restraining the growth of the facial erythema treatment market. The opportunities available in the market such as increasing advancement in drug product, rising R&D activities.
New Product launch to flourish the market:
Major Key market manufacturer have been focusing on producing new product solution for specific applications as there are varying demands based on the utilization. Leading market players have determined how improved accuracy can be influential in many applications. In 2019, The US FDA has approved the “Rhofade, Allergan” (oxymetazoline hydrochloride cream) it is the first alpha1A adrenoceptor agonist which is used for the cure of the persistent facial erythema generally related with the rosacea.
Key benefits of the report:
- This study presents the analytical depiction of the global facial erythema treatment market along with the current trends and future estimations to determine the imminent investment pockets.
- The report presents information related to key drivers, restraints, and opportunities along with detailed analysis of the global facial erythema treatment market share.
- The current market is quantitatively analyzed to highlight the global facial erythema treatment market growth scenario.
- Porter’s five forces analysis illustrates the potency of buyers & suppliers in the market.
- The report provides a detailed global facial erythema treatment market analysis based on competitive intensity and how the competition will take shape in coming years.
Questions answered in the Facial Erythema Treatment Market research report:
- Who are the leading market players active in facial erythema treatment market?
- What are the current trends will influence the market in the next few years?
- What are the driving factors, restraints, and opportunities in the market?
- What are the projections for the future that will help in taking further strategic steps?
Facial Erythema Treatment Market Report Highlights
| Aspects | Details |
| By Drug Type |
|
| By Route of Administration |
|
| By Disease Type |
|
| By Distribution Channel |
|
| By Region |
|
| Key Market Players | Anacor Pharmaceuticals Inc., Pfizer Inc., Novartis International AG, Sanofi S.A., Valeant Pharmaceuticals International, Inc., Astellas Pharma Inc., Meda Pharmaceuticals, GlaxoSmithKline plc,, Regeneron Pharmaceuticals Inc., Galderma S.A., Bausch Health Companies Inc. |
Loading Table Of Content...