Fibrinogen Concentrate Market Research, 2033
The global fibrinogen concentrate market size was valued at $3.3 billion in 2023, and is projected to reach $5.9 billion by 2033, growing at a CAGR of 6.1% from 2024 to 2033. The fibrinogen concentrate market growth is influenced by various factors that drive, restrain, and create opportunities for its growth.
Market Introduction and Definition
Fibrinogen concentrate is a plasma-derived product used to manage and prevent bleeding. It is used for patients with congenital or acquired fibrinogen deficiency. When fibrinogen levels are insufficient or dysfunctional, patients are at high risk of excessive or uncontrolled bleeding. Fibrinogen concentrate is a soluble glycoprotein synthesized in the liver. It plays an important role in the final stages of the coagulation cascade.
Rapid correction of low fibrinogen levels play an important role in emergency and critical care settings. This helps prevent life-threatening hemorrhage. Fibrinogen concentrate offers a targeted and efficient method of replacement therapy compared to traditional blood components.
Commercially available fibrinogen concentrates can be administered intravenously. This allows for precise dosing based on body weight and plasma fibrinogen levels. Further, fibrinogen concentrate is available and does not require blood type matching. This makes it especially valuable in emergency situations, where time is critical. In surgical settings, fibrinogen concentrate is used as part of patient blood management programs. Rapid supplementation with fibrinogen concentrate can stabilize clot formation and reduce transfusion requirements. Fibrinogen concentrate also plays an important role in restoring adequate clot strength when fibrinogen levels fall below critical thresholds.
Fibrinogen concentrate is a therapeutic product derived from human plasma or recombinant technology, used to treat bleeding disorders. The making procedure involves the collection of plasma, followed by purification and concentration of fibrinogen. For recombinant fibrinogen, genetic engineering techniques are used to produce fibrinogen in cell cultures. Applications of fibrinogen concentrate include treating congenital fibrinogen deficiencies (like afibrinogenemia and hypofibrinogenemia) , managing acute bleeding episodes in surgical and trauma patients, and promoting blood clotting in various medical procedures. Its ability to restore fibrinogen levels rapidly makes it essential for critical care and surgical settings. Advancements in biotechnology are also driving the development of recombinant fibrinogen products. These advancements aim to reduce reliance on human plasma and further improve safety profiles.
Key Takeaways
- The fibrinogen concentrate market share study covers 20 countries. The research includes a segment analysis of each country in terms of value ($Billion) for the projected fibrinogen concentrate market forecast period 2024-2033.
- More than 1, 500 product literatures, industry releases, annual reports, and other such documents of major fibrinogen concentrate industry participants along with authentic industry journals, trade associations' releases, and government websites have been reviewed for generating high-value industry insights.
- The study integrated high-quality data, professional opinions and analysis, and critical independent perspectives. The research approach is intended to provide a balanced view of global markets and to assist stakeholders in making educated decisions in order to achieve their most ambitious growth objectives.
Key Market Dynamics
Increase in global volume of complex surgical procedures drive the growth of the fibrinogen concentrate market. The shift toward patient blood management programs further boosts the demand for fibrinogen concentrate. Surge in incidence of trauma and road accidents also fuels the market growth. In addition, rise in awareness of postpartum hemorrhage management and improvements in maternal healthcare infrastructure also contribute to increased adoption rates.
Advancements in point-of-care diagnostic tools make way for targeted use of fibrinogen concentrate. These tools enable clinicians to assess fibrinogen function in real time. This supports evidence-based decision-making and improves patient outcomes.
High cost of fibrinogen concentrate compared to traditional therapies limit the growth of the market. Clinicians continue to rely on conventional blood components due to familiarity or cost considerations. This also deters the market growth.
Improvements in healthcare infrastructure and rise in incidences of trauma surge the demand for advanced hemostatic therapies. Expansion of indications for acquired bleeding conditions presents another growth avenue. The development of recombinant fibrinogen products offers a promising opportunity to address supply limitations and enhance safety. Ongoing clinical research supporting early fibrinogen replacement in trauma and obstetric care is expected to expand clinical acceptance. Strategic collaborations between manufacturers and healthcare institutions, along with investments in education and training programs, can further drive adoption. As precision medicine and goal-directed therapy gain momentum, fibrinogen concentrate is poised to play an increasingly central role in modern coagulation management.
What Are the Strategies Adopted by the Key Players in the Market?
Product development and launch are the key strategies adopted by the key players of the market. Many companies focus on improving their product portfolios with new formulations, including ready-to-use liquid fibrinogen concentrates or enhanced lyophilized products, to meet clinical needs more effectively and support faster hemostasis in acute bleeding situations. This emphasis on innovation helps differentiate offerings in a market where clinical efficacy and safety are paramount.
Another common strategy is geographic expansion and distributor partnerships. Several firms, particularly Western multinationals, have been partnering with regional distributors or local healthcare providers, especially in Asia-Pacific and Latin America, to increase product accessibility and address untapped demand. These collaborations often include localized manufacturing agreements or distribution agreements tailored to regional regulatory requirements.
Regulatory approval efforts also play a strategic role. Obtaining clearances from key regulatory bodies such as the U.S. FDA and the European Medicines Agency (EMA) allows companies to expand indications or secure label updates, supporting broader clinical use and adoption among healthcare providers. Firms invest in clinical trials to generate data that strengthens product positioning.
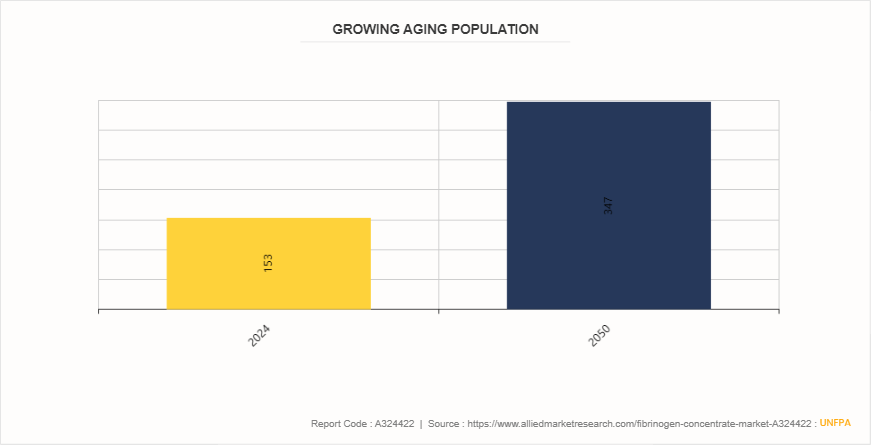
Growing Aging Population for Fibrinogen Concentrate Market
According to the United Nations Population Fund report from December 2023, the elderly population (aged 60 and above) is projected to rise from 153 million to 347 million by 2050. This significant demographic shift represents a profound societal transformation with extensive implications, particularly in healthcare. As the elderly population grows, so does the prevalence of age-related bleeding disorders and surgical needs, driving the demand for fibrinogen concentrate. Hospitals and healthcare facilities increasingly rely on fibrinogen concentrate to manage bleeding complications in older patients, making it a crucial component in improving health outcomes and quality of life for this expanding age group.
Market Segmentation
The fibrinogen concentrate industry is segmented into product type, application, end user and region. On the basis of product type, the market is categorized into human fibrinogen concentrate, recombinant fibrinogen concentrates, animal-derived fibrinogen concentrate, and synthetic fibrinogen concentrate. As per application, the market is divided into congenital fibrinogen deficiency, surgical procedures, and other applications. On the basis of end user, the market is segmented into hospitals, ambulatory surgical centers, and others. Region wise, it is analyzed across North America, Europe, Asia-Pacific, and LAMEA.
Regional/Country Market Outlook
The fibrinogen concentrate market share varies significantly by region. In North America and Europe, high adoption rates are driven by advanced healthcare infrastructure, strong regulatory frameworks, and a high prevalence of bleeding disorders. These regions benefit from well-established plasma collection and processing facilities. In Asia-Pacific, increasing healthcare investments and rising surgical procedures drive market growth, although cost and access issues remain. Latin America and the Middle East & Africa show growing interest due to expanding healthcare systems and rising awareness, but market penetration is constrained by higher costs and limited access to advanced treatments. Each region presents unique opportunities and challenges based on local healthcare dynamics.
How Intense Is Market Competition and What Defines Competitive Advantage?
Major companies control large portions of global sales, especially in North America and Europe, making market penetration by newcomers more challenging due to the scale and resource advantages of incumbents. Complex regulatory pathways for plasma-derived and recombinant products raise entry costs, requiring substantial investment in clinical evidence and manufacturing standards. New entrants must clear these hurdles to gain market access. Competitive advantage increasingly shifts toward product innovation rather than price alone. Companies investing in next-generation formulations or recombinant technologies gain differentiation by offering improved safety, potency, and ease of use. Products from established manufacturers benefit from clinician familiarity and inclusion in clinical guidelines, supporting adoption in hospitals and surgical centers. This trust reinforces competitive positioning.
What Makes the U.S., Europe, and Asia-Pacific Key Markets for Industry Players?
United States (U.S.)
The United States is a core market due to its large healthcare system, high surgical volumes, and advanced trauma care networks. As part of North America’s overall dominance, the U.S. accounts for a significant share of global fibrinogen concentrate demand. Its well-developed regulatory environment and strong R&D ecosystem attract major global players who invest in both product approval and clinical evidence generation. High prevalence of bleeding disorders and growing surgical procedures drive steady demand for fibrinogen therapy.
Europe
Europe remains a key industry market due to its extensive healthcare infrastructure and coordinated regulatory frameworks across many nations. The European Medicines Agency (EMA) supports innovation while ensuring product safety, which encourages adoption in surgical and emergency care settings. Countries such as Germany, France, and the UK have influential healthcare systems where fibrinogen concentrates are increasingly integrated into blood management protocols. Regional clinical trials, strong plasma fractionation capacity, and established reimbursement policies further strengthen Europe’s position.
Asia-Pacific
Asia-Pacific is an opportunistic market industry players due to rapid expansion of healthcare infrastructure and increase in awareness of coagulation disorders. Countries such as China, India, Japan, and South Korea are investing in healthcare modernization, which boosts demand for fibrinogen products. The presence of aging population creates a growing patient base for treatable bleeding conditions. Local manufacturers and improved plasma networks support market expansion, making Asia-Pacific crucial for future industry growth.
Industry Trends
- In April 2022, Grifols, a global leader in plasma-derived medicines helping to improve people's health and well-being for more than 110 years, announced the completion of the acquisition of 100% of the share capital of Tiancheng (Germany) Pharmaceutical Holdings AG, a German company that holds 89.88% of the ordinary shares and 1.08% of the preferred shares of Biotest AG, a European healthcare company specialized in innovative hematology and clinical immunology.
Competitive Landscape
The major players operating in the fibrinogen concentrate market include Baxter International Inc, CSL Behring, Enzo Life Sciences, Inc, Ethicon (Johnson and Johnson) , GC Biopharma, Hualan Biological Engineering Inc., LFB, Molecular Innovations Inc, Octapharma AG, and ProFibrix BV. Other players in the fibrinogen concentrate market size include Shanghai RAAS, ThermoFisher Scientific, and so on.
Which Region Dominates the Global Market and What Drives Its Leadership?
The North America region currently dominates the global fibrinogen concentrate market and is expected to maintain its leadership position through the forecast period. According to Allied Market Research, North America held the largest share of revenue in 2023 and continues to outperform other regions in both adoption and overall clinical utilization.
The United States accounts for a significant portion of this dominance due to its advanced healthcare infrastructure, widespread access to specialized medical care, and well-developed hospital systems.
In addition to surgical demand, North America has a relatively high prevalence of chronic and bleeding disorders, including congenital fibrinogen deficiencies and coagulopathies, which increases clinical demand for reliable coagulation management products. The presence of key market players and robust investment in research and development supports a continuous flow of product innovations, further strengthening the region’s competitive edge.
Regulatory approval processes in the region also favor early product launches. With clear guidelines from agencies such as the U.S. FDA, manufacturers can achieve timely market entry, which supports extensive clinical uptake across hospitals and trauma centers.
Well-established reimbursement frameworks in both public and private healthcare systems help ensure that fibrinogen concentrate products are more affordable and accessible for patients and healthcare providers. Overall, strong healthcare spending, advanced clinical practices, and solid regulatory support make North America the leading region in the fibrinogen concentrate market.
How Do Regulatory Frameworks Influence Market Development?
Regulatory frameworks play a critical role in shaping the development, commercialization, and adoption of fibrinogen concentrate globally. As a plasma-derived or recombinant biologic therapy, fibrinogen concentrate is subject to strict oversight to ensure product safety, quality, and clinical efficacy. According to insights from Allied Market Research, regulatory clarity and supportive approval pathways are key factors driving growth in developed regions, while complex or evolving regulations can slow expansion in emerging markets.
In major markets such as the United States, regulatory oversight by the U.S. Food and Drug Administration (FDA) significantly influences product development timelines. Manufacturers must comply with stringent requirements related to plasma sourcing, viral inactivation, manufacturing standards, and clinical trial data.
Similarly, in Europe, regulation by the European Medicines Agency (EMA) ensures regulations across EU member states. EMA approval facilitates access to multiple countries under a centralized framework, enabling manufacturers to scale distribution more efficiently.
Regulatory frameworks also influence clinical indications and labeling. Initially, fibrinogen concentrate approvals focused primarily on congenital fibrinogen deficiency.
In emerging markets across Asia-Pacific and Latin America, regulatory systems are evolving. Some countries are streamlining biologics approval processes to attract foreign investment and improve access to advanced therapies.
Recent Key Strategies and Developments
- In March 2021, Octapharma received the U.S. Food and Drug Administration (FDA) approval for two supplements related to Biological License Applications, thus strengthening the company's pediatric critical care product portfolio. The supplement approvals resulted in new product labeling for Octaplas, Pooled Plasma (Human) , and Fibryga, Fibrinogen (Human) Lyophilized Powder for Reconstitution.?
Key Sources Referred
- World Health Organization (WHO)
- Centers for Medicare & Medicaid Services (CMS)
- National Health Service (NHS)
- National Health Mission (NHM)
- Octapharma
- Institute for Health Metrics and Evaluation
- Centers for Disease Control and Prevention (CDC)
- Food and Drug Administration (FDA)
- National Institutes of Health (NIH)
Key Benefits for Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the fibrinogen concentrate market analysis from 2024 to 2033 to identify the prevailing fibrinogen concentrate market opportunity.
- The market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the fibrinogen concentrate industry segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global fibrinogen concentrate market trends, key players, market segments, application areas, and market growth strategies.
Fibrinogen Concentrate Market , by Product Type Report Highlights
| Aspects | Details |
| Market Size By 2033 | USD 5.9 Billion |
| Growth Rate | CAGR of 6.1% |
| Forecast period | 2024 - 2033 |
| Report Pages | 216 |
| By Product Type |
|
| By Application |
|
| By End User |
|
| By Region |
|
| Key Market Players | Johnson and Johnson, Hualan Biological Engineering Inc., Octapharma AG, Shanghai RAAS, ProFibrix BV., Enzo Life Sciences, Inc, ThermoFisher Scientific, GC Biopharma, CSL Behring, Baxter International Inc |
The total market value of fibrinogen concentrate market was $3.3 billion in 2023.
The forecast period for fibrinogen concentrate market is 2024 to 2033
The market value of fibrinogen concentrate market is projected to reach $5.9 billion by 2035.
The base year is 2023 in Fibrinogen Concentrate market .
Fibrinogen concentrate is a therapeutic product derived from human plasma or recombinant technology, used to treat bleeding disorders. The making procedure involves the collection of plasma, followed by purification and concentration of fibrinogen.
Loading Table Of Content...



