Foot And Mouth Disease (FMD) Vaccines Market Research, 2033
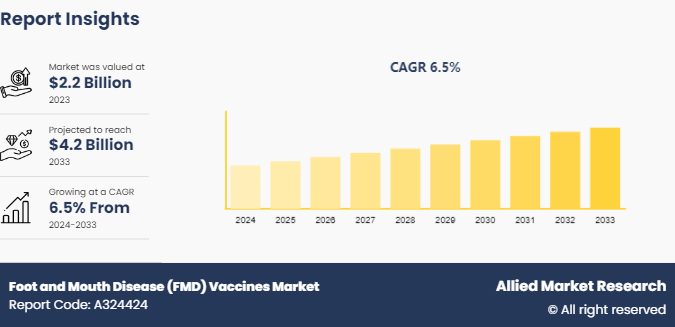
The global foot and mouth disease vaccines market size was valued at $2.2 billion in 2023, and is projected to reach $4.2 billion by 2033, growing at a CAGR of 6.5% from 2024 to 2033. The rise in awareness about the economic impact of foot and mouth disease and the benefits of the vaccination are driving the demand for innovative solutions in the foot and mouth disease vaccines market.
Market Introduction and Definition
Foot and mouth disease is a contagious viral disease that affects cattle, sheep, goats, and pigs. Symptoms include fever, lameness, and vesicular lesions in the mouth and on the feet. Vaccination is one of the major important tools to control and prevent outbreaks. These vaccines are designed to stimulate protective immunity without causing disease.
Vaccine banks are often established at national or regional levels to ensure rapid access to appropriate vaccine strains during outbreaks. High-potency vaccines are important in emergency vaccination campaigns to build herd immunity and limit virus spread. The duration of immunity varies depending on vaccine formulation, animal species, and management practices, often necessitating biannual or annual revaccination in endemic regions.
Foot and mouth disease vaccines market are biological preparations designed to immunize livestock against the highly contagious virus, which affects cloven-hoofed animals. These vaccines contain inactivated or attenuated virus particles that stimulate the immune system of the animal to recognize and fight the actual virus. Upon vaccination, the immune system produces specific antibodies that neutralize the virus, preventing it from causing the disease. Booster doses are often required to maintain immunity. By enhancing herd immunity, these vaccines play a crucial role in controlling outbreaks, minimizing economic losses, and promoting animal health in affected regions.
Traditional inactivated foot and mouth disease vaccines have limitations. They require high-biosafety production facilities because large quantities of live virus must be handled prior to inactivation. There is a small but significant risk associated with incomplete inactivation if manufacturing standards are not strictly maintained. Furthermore, vaccinated animals cannot always be easily distinguished from naturally infected animals using conventional serological tests, complicating trade and disease surveillance. To address this, marker vaccines and companion diagnostic tests have been developed, targeting non-structural proteins absent in purified vaccines.
Advancements in biotechnology make way for next-generation foot and mouth disease vaccines. Recombinant protein vaccines, viral vector vaccines, and peptide-based vaccines are under development to improve safety and allow DIVA compatibility. Some research also focuses on thermostable vaccine formulations to reduce reliance on cold chain logistics, particularly in tropical and resource-limited settings.
Key Takeaways
- The foot and mouth disease vaccines market share study covers 20 countries. The research includes a segment analysis of each country in terms of value for the projected foot and mouth disease vaccines market forecast period.
- More than 1, 500 product literatures, industry releases, annual reports, and other such documents of major foot and mouth disease vaccines industry participants along with authentic industry journals, trade associations' releases, and government websites have been reviewed for generating high-value industry insights.
- The study integrated high-quality data, professional opinions and analysis, and critical independent perspectives. The research approach is intended to provide a balanced view of global markets and to assist stakeholders in making educated decisions in order to achieve their most ambitious growth objectives.
Key Market Dynamics
High economic impact of foot and mouth disease outbreaks drive the growth of the market. The disease can lead to massive production losses due to decrease in milk yield and weight loss. Countries heavily dependent on livestock exports are particularly motivated to invest in vaccination programs to protect their market access and maintain foot and mouth disease-free status. Rise in global demand for meat and dairy products is a major factor that drives the growth of the market. Governments and producers are therefore prioritizing preventive healthcare, including vaccination, to secure stable supply chains.
Government-supported vaccination campaigns and international collaborations also drive vaccine demand. Further, increase in awareness of biosecurity and improved veterinary infrastructure in developing countries contribute to higher vaccine uptake. Public-private partnerships are also facilitating technology transfer and local production capabilities.
A major challenge is antigenic diversity and frequent viral mutation. Continuous monitoring and vaccine strain updates are necessary, increasing production complexity and cost. Cold chain requirements represent another significant barrier, particularly in remote or resource-limited areas. Inactivated vaccines generally require refrigeration, and disruptions can compromise potency. Logistical challenges reduce coverage rates and program efficiency. Trade-related concerns present an additional restraint. Some FMD-free countries prefer not to vaccinate routinely because vaccination may complicate serological surveillance and impact export status. This policy choice can reduce consistent vaccine demand in certain regions.
Development of next-generation vaccines is one of the major lucrative growth opportunities in the market. Recombinant and virus-like particle technologies offer safer production without handling live FMD virus, potentially reducing costs and biosafety risks. DIVA-compatible vaccines present strong commercial potential, as they facilitate disease monitoring and international trade compliance. Thermostable vaccine formulations represent another promising area, especially for tropical climates. Improved stability could expand vaccine coverage in underserved regions and reduce wastage.
Number of Bovines according to Livestock Census for Foot and Mouth Disease (FMD) Vaccines Market
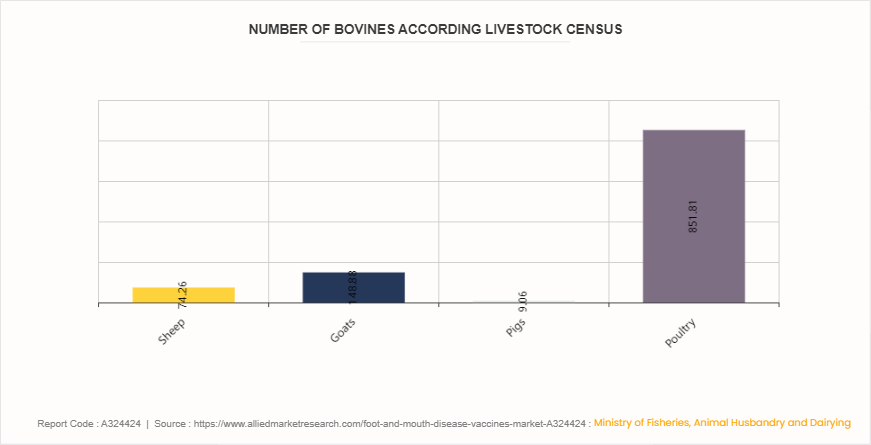
According to the 2023 report by the Ministry of Fisheries, Animal Husbandry and Dairying, India hosts approximately 303.76 million bovines, 74.26 million sheep, 148.88 million goats, 9.06 million pigs, and 851.81 million poultry as per the 20th Livestock Census. The vast livestock population underscores the critical need for foot and mouth disease (FMD) vaccines to protect these animals from the highly contagious and economically devastating FMD. Effective vaccination programs are essential to maintain animal health, ensure agricultural productivity, and safeguard the livelihoods of millions dependent on livestock farming in the country.
Market Segmentation
The foot and mouth disease vaccines industry is segmented into product type, animal type, and region. On the basis of product type, the market is bifurcated into conventional vaccines, and emergency vaccines. On the basis of animal type, the market is categorized into cattle, sheep and goat, pig, and others. Region-wise, it is analyzed across North America, Europe, Asia-Pacific, and LAMEA.
Which Segment Holds the Largest Market Share and Why?
In the foot and mouth disease vaccines market, by product type, the inactivated (conventional) vaccine segment holds the largest share. Inactivated vaccines are well-established and are widely approved by government and veterinary authorities for routine preventive immunization. This drives the dominance of the segment. By animal type, the cattle vaccines segment accounts for over half of global vaccine usage. Cattle are highly susceptible to foot and mouth disease and incur significant economic losses when infected, especially in beef and dairy industries. Many countries mandate routine vaccination for cattle, boosting demand. By distribution channel, the veterinary hospitals and clinics segment boost the growth of the market. These facilities serve as the primary point of care delivery and trusted vaccination providers for livestock producers.
Regional/Country Market Outlook
The FMD Vaccines Market Sizevaries significantly by region. In Asia-Pacific and Africa, where FMD is endemic, there is a high demand for vaccines due to frequent outbreaks, driving the market growth. Latin America also sees substantial demand, particularly in countries with large cattle industries. Europe and North America, with stringent biosecurity measures and fewer outbreaks, have moderate demand but invest heavily in preventive vaccination programs. Emerging economies, particularly in Asia-Pacific, are expected to witness rapid growth in the foot and mouth disease (FMD) vaccines market due to increase in livestock populations and government initiatives to control the disease.
- On January 1, 2024, according to American Farm Bureau Federation, all cattle and calves in the U.S., were 87.2 million head, 2% lower than in 2023.
Where Are the Most Attractive Investment Opportunities in the FMD Vaccines Market?
One of the most attractive investment opportunities lies in next-generation vaccine technologies. Traditional inactivated vaccines currently dominate the market, but innovations such as recombinant vaccines, multivalent formulations, thermostable formats, and marker vaccines (DIVA) are gaining traction. These advanced vaccines can address limitations of conventional products by offering broader protection, reduced cold-chain dependence, longer immunity, and easier disease surveillance, which is especially valuable in resource-limited regions.
Geographical expansion represents another important investment opportunity. The Asia-Pacific region continues to lead in demand due to its large and rapidly growing livestock populations and frequent FMD outbreaks. Governments in countries such as China, India, and Indonesia have implemented extensive vaccination programs, supported by regulatory frameworks and public funding. The rising livestock sector and increasing meat and dairy consumption in these markets signal sustained vaccine demand.
Additionally, investments in cold-chain infrastructure and distribution platforms are critical. Efficient storage and delivery systems help extend vaccine shelf life, reduce wastage, and support mass immunization campaigns. Given the significant logistical challenges in many developing regions, companies that build or invest in reliable distribution networks are poised to capture long-term demand.
Industry Trends
- According to the Ministry of Fisheries, Animal Husbandry and Dairying report of 2023, 12.61 and 1.80 crore animals are vaccinated for Round III and Round IV, respectively, of FMD vaccination.
Competitive Landscape
The major players operating in the foot and mouth disease (FMD) vaccines market include Biogenesis Bago, Boehringer Ingelheim GmbH, Brilliant Bio Pharma Pvt Ltd., China Animal Husbandry Group, Indian Immunologicals Limited, Limor de Colombia, Merck & Co. Inc., VECOL S.A, VETAL Animal Health Products, and Biovet. Other key players include Ceva Sante Animale, Veterinary Serum and Vaccine Research Institute, and so on.
What Strategies Should New Entrants Adopt to Succeed in This Market?
Entering the Foot and Mouth Disease (FMD) vaccines market requires well-planned strategies due to its regulated nature, technical complexity, and competitive landscape. New entrants should focus on a combination of innovation, partnerships, compliance, and market differentiation to establish a strong presence.
New companies need to invest in research and development. The market is shifting from conventional inactivated vaccines toward advanced formulations such as recombinant, multivalent, thermostable, and DIVA-compatible vaccines. These next-generation products meet emerging demand for broader protection, easier logistics, and improved disease surveillance—factors that are increasingly valued by governments and large livestock producers. Prioritizing R&D enables entrants to offer competitive products that fill unmet needs and differentiate from existing offerings.
Also, collaboration with local and international partners is essential. Forming public-private partnerships (PPPs) with government veterinary departments, regional vaccine banks, and international health organizations can facilitate market entry and improve access to public vaccination programs. PPPs reduce financial risk, align with policy goals, and expand distribution networks, making it easier for new players to gain market share.
How Can Businesses Strengthen Market Position and Expand Globally?
Innovation and Product Diversification should remain central to strengthening market position. Investing in next-generation vaccines—such as recombinant, multivalent, thermostable, and DIVA-compatible formats—can give companies an edge over competitors reliant solely on traditional inactivated vaccines. These advanced products offer enhanced protection, simpler logistics, and compatibility with global disease surveillance standards, making them attractive to both endemic and FMD-free regions looking to maintain herd immunity. Diversifying product portfolios also reduces reliance on single product lines and opens opportunities in specialized segments. Geographic expansion into high-growth regions is another key strategy. The Asia-Pacific market represents a substantial share of global demand due to large livestock populations and frequent outbreaks, especially in China, India, and Southeast Asian countries. Establishing regional manufacturing and distribution hubs in these areas can reduce lead times, lower transportation costs, and improve responsiveness during outbreaks. Tailoring products and marketing strategies to local needs accelerates adoption and strengthens market presence.
Strengthening distribution networks is essential for global coverage. Partnering with veterinary clinics, agricultural cooperatives, government procurement offices, and regional distributors ensures vaccines reach end users efficiently. Investing in cold-chain infrastructure and logistics further enhances reliability and reduces wastage, particularly in rural and underserved regions where logistical challenges often limit vaccine access. Collaborative initiatives with governments, non-governmental organizations, and global health bodies can also elevate market positioning. By participating in mass vaccination drives, disease surveillance programs, and emergency response frameworks, companies can align with public health goals and position themselves as trusted partners. Engaging in public-private partnerships (PPPs) leads to shared investment risks, broader distribution channels, and increased public sector demand.
Recent Key Strategies and Developments
- In July 2020, Zoetis, Inc., the largest global animal health company, announced the application for a petition to the Animal and Plant Health Inspection Services to approve the manufacture of foot and mouth disease vaccine in the U.S.
Key Sources Referred
- World Health Organization (WHO)
- Centers for Medicare & Medicaid Services (CMS)
- National Health Service (NHS)
- Ministry of Fisheries, Animal Husbandry & Dairying
- National Health Mission (NHM)
- Institute for Health Metrics and Evaluation
- Centers for Disease Control and Prevention (CDC)
- Food and Drug Administration (FDA)
- National Institutes of Health (NIH)
Key Benefits for Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the Foot and Mouth Disease Vaccines Market Analysis from 2024 to 2033 to identify the prevailing Foot and Mouth Disease Vaccines Market opportunity
- The market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the foot and mouth disease (FMD) vaccines market segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global Foot and Mouth Disease Vaccines Market Trends, key players, market segments, application areas, and market growth strategies.
Foot and Mouth Disease Report Highlights
| Aspects | Details |
| Market Size By 2033 | USD 4.2 Billion |
| Growth Rate | CAGR of 6.5% |
| Forecast period | 2024 - 2033 |
| Report Pages | 216 |
| By Product Type |
|
| By Animal Type |
|
| By Region |
|
| Key Market Players | VETAL Animal Health Products, VECOL S.A, China Animal Husbandry Group, Biogenesis Bago, Brilliant Bio Pharma Pvt Ltd, Limor de Colombia, Boehringer Ingelheim GmbH, Biovet, Merck & Co. Inc., Indian Immunologicals Limited |
The total market value of foot and mouth disease vaccines market was $2.2 billion in 2023.
The forecast period for foot and mouth disease vaccines market is 2024 to 2033
The market value of foot and mouth disease vaccines market is projected to reach $ 4.2 billion by 2033
The base year is 2023 in foot and mouth disease vaccines market .
Foot and mouth disease (FMD) vaccines are biological preparations designed to immunize livestock against the highly contagious FMD virus, which affects cloven-hoofed animals.
Loading Table Of Content...



