Formulation Development Outsourcing Market Research, 2031
The global formulation development outsourcing market size was valued at $22.5 billion in 2021, and is projected to reach $51.9 billion by 2031, growing at a CAGR of 9% from 2022 to 2031. The formulation development outsourcing market demand is growing rapidly owing to increasing number of clinical trials and regulatory requirements. The need for specialized knowledge in formulation development and regulatory compliance can be met through outsourcing, as these service providers have extensive experience and expertise in the field.

The formulation development outsourcing market size is driven by increasing demand for biopharmaceuticals, advanced drug delivery systems, and complex dosage forms has further fueled the growth of the formulation development outsourcing market. These advanced drug delivery systems require specialized formulation development, which can be met through outsourcing, leading to the growth of the market. The increased demand for cost-effective drug development, the need for specialized expertise, and the growing number of clinical trials and regulatory requirements are the key drivers of the formulation development outsourcing market.
Drug development is a time-consuming, costly, and complex process that is full of uncertainty about whether or not a drug will be successful. Every stage of drug development needs money. Long development timeframes and low success rates result in expensive R&D expenses overall. The most significant challenges restricting market growth are structural changes in the pharmaceutical sector and a lack of funds to complete the medication development process through formulation development.
Chronic diseases and conditions are becoming more common all over the world. Europe's pharmaceutical and biotechnology businesses have boosted their R&D spending in recent years in response to the growing demand for novel drugs. This might be attributed to an aging population, chronic illnesses, and infectious diseases. The creation of anti-cancer pharmaceutical formulations is crucial since formulation includes stability, solubility, and bioavailability throughout drug development.
Global awareness of vaccines is leading to a surge in outsourcing formulation development services. Also, more and more pharma/biopharma companies are planning to partner with Contract Development & Manufacturing Organization (CDMOs), much of this activity is occurring in the early phase of development with the goal of overcoming risk, along with saving time and money as a drug passes through the development pipeline.
The key players profiled in this report include Charles River Laboratories. Syngene International Limited, Catalent, Inc, Piramal Pharma Ltd., Intertek Group plc, Thermo Fisher Scientific, Eurofins Scientific, Laboratory Corporation of America Holdings., Dr. Reddy’s Laboratories Ltd., and EMERGENT.
The global formulation development outsourcing market is segmented on the basis of service type, route of formulation, end user, and region. By service type, the market is segmented into pre-formulation, formulation development. By route of formulation, the market is classified into oral, injectable, and others. By end user, the market is segmented into pharmaceutical industries, research and academic institutes. By region, the market is analyzed across North America, Europe, Asia-Pacific, and LAMEA.
The formulation development outsourcing market is segmented into Services Type, Route of Formulation and End-user.
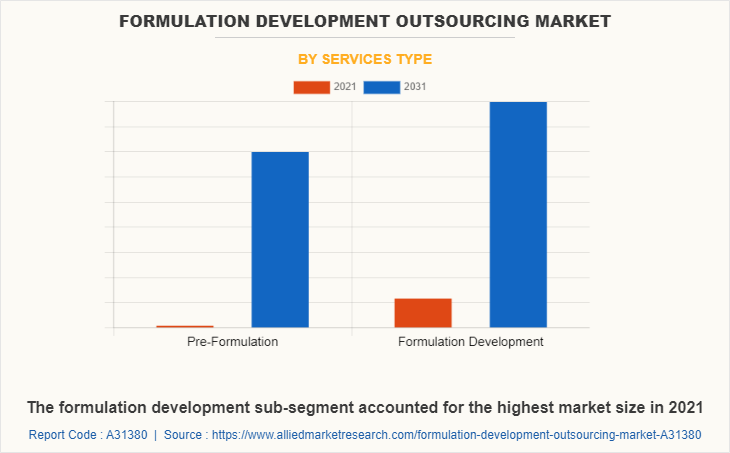
By services type, the pre-formulation segment dominated the market in 2021. Pre formulation helps researchers narrow down potential leads by evaluating their physicochemical and biological characteristics. Every stage of clinical trials requires the development of a formulation, and the number of clinical research has greatly expanded over time. The market for outsourcing formulation development is projected to benefit from the increasing number of clinical studies. Such factor is expected to drive the market share in the forecast time period.
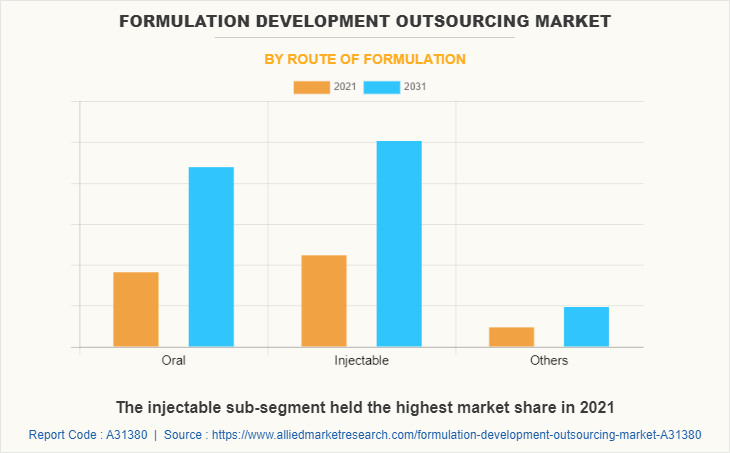
By route of formulation, the injectable segment dominated the global formulation development outsourcing market share in 2021. Injectables, also known as parenteral preparations, are sterile pharmaceuticals that are injected directly into a person's body. Solutions, suspensions, emulsions for injection or infusion, powders for injection or infusion, gels for injection, and implants are examples of parenteral preparations. Long-acting injectable formulations are widely used for contraception, the treatment of immunodeficiencies and psychiatric disorders, as well as for adherence to medication in general. This factor is likely to propel market growth in the anticipated time period.
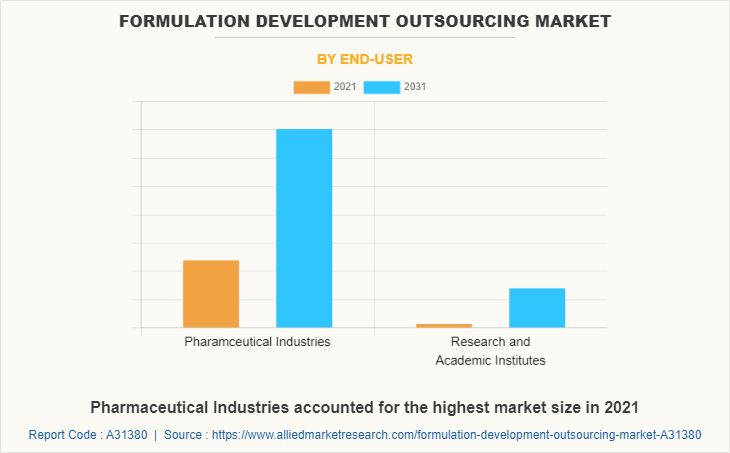
By end-user, the pharmaceutical industries segment dominated the global formulation development outsourcing market share in 2021. Rapidly increasing cases of chronic illness among people and rising demand for potent drugs or vaccine has boosted the pharmaceutical companies to initiate their research and development activities and develop new drugs. Many pharma companies, already have drugs in their pipelines and at different clinical stages. Thus, high demand from pharmaceutical companies for new drug and vaccine development is expected to drive the formulation development outsourcing market in the forecast time period.
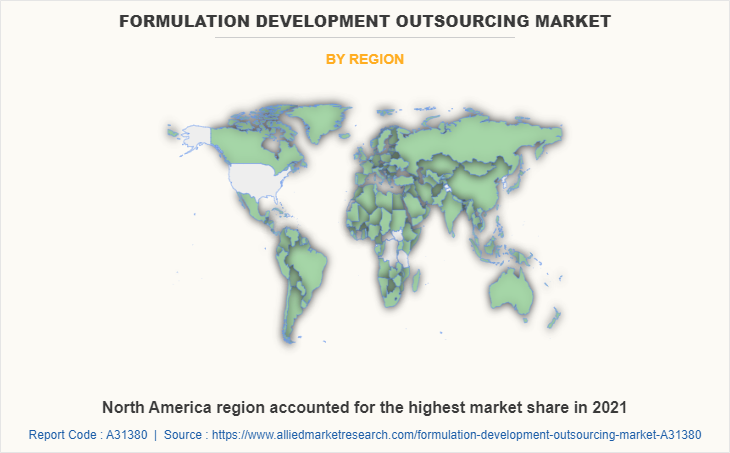
By region, North America dominated the global market in 2021 and is projected to remain the fastest-growing segment during the forecast period. The U.S. formulation development market is anticipated to have substantial expansion in the research industry due to the increased prevalence of chronic diseases and high demand for new and effective drugs to treat these diseases. Also, number of clinical trials for drug development in the region is extremely high.
Impact of COVID-19 on the Global Formulation Development Outsourcing Industry
- The global economy was impacted by the COVID-19, and especially the pharmaceutical industry. The largest economic catastrophe in modern history has also been compounded by the health disaster
- The COVID-19 pandemic has a huge impact on the formulation development outsourcing business as nations' health systems engaged quickly in research and development to combat the virus Manufacturing companies and research agencies either worked alone or collaborated together to find and develop potential treatment drug for the novel coronavirus infection
Highlights of the Report
- The report provides an exclusive and comprehensive analysis of the global formulation development outsourcing market trends along with the formulation development outsourcing market forecast
- The report elucidates the formulation development outsourcing market opportunity along with key drivers and restraints of the market. It is a compilation of detailed information and inputs from industry participants and industry experts across the value chain, as well as quantitative and qualitative assessment by industry analysts
- Porter’s five forces analysis helps analyse the potential of the buyers & suppliers and the competitive scenario of the market for strategy building
- The report entailing the formulation development outsourcing market analysis maps the qualitative sway of various industry factors on market segments as well as geographies
- The data in this report aims on market dynamics, trends, and developments affecting the formulation development outsourcing market growth
Formulation Development Outsourcing Market Report Highlights
| Aspects | Details |
| Market Size By 2031 | USD 51.9 billion |
| Growth Rate | CAGR of 9% |
| Forecast period | 2021 - 2031 |
| Report Pages | 240 |
| By Services Type |
|
| By Route of Formulation |
|
| By End-user |
|
| By Region |
|
| Key Market Players | Laboratory Corporation of America Holdings, Emergent BioSolutions, Inc., syngene international limited, Eurofins Scientific, Piramal Pharma Limited, Thermo Fisher Scientific Inc., Intertek Group Plc, Dr Reddy's Laboratories Ltd., Catalent Inc, Charles River Laboratories International, Inc. |
Analyst Review
Pharmaceutical corporations are moderately outsourcing R&D activities to academia and private contract research organizations (CROs) to reduce drug development schedules and costs. According to an article published in 2018 titled "The Era of Pharma and Biotech Outsourcing," an estimated 75.0-80.0% of R&D investment in the biopharmaceutical sector can be outsourced, offering the potential for CROs. This, in turn, may enhance the market growth. The expanding number of end-to-end service providers concentrating on meeting the growing need for lower-cost medication development and manufacture is likely to boost market expansion. Another important issue limiting market development is structural changes in the pharmaceutical business. Several developments have occurred in the global pharmaceutical sector during the previous decade. Such developments have put pressure on major pharmaceutical transnational companies (TNCs) dependent on research and development (R&D) to adapt the way they do business. Other pressures, like regulatory and antitrust difficulties, have also been applied to major R&D-based TNCs. Regulatory barriers have grown, with drug regulatory bodies asking more questions about clinical data presented for prospective blockbusters. The formulation development outsourcing market is predicted to rise steadily in the future years, owing to the market's rising need for the creation of innovative treatments. As major pharmaceutical and biopharmaceutical companies have increased their focus on the development of numerous therapeutics aimed at providing effective treatment for a wide range of chronic diseases, the demand for formulation development services is expected to rise in the future, as outsourcing is a viable option for these companies. With blockbuster pharmaceuticals nearing patent expiration, corporations are likely to strive for the creation of novel treatments, which will gain impetus for the outsourcing of formulation development.
Among the analyzed regions, the Asia-Pacific region is expected to account for the highest revenue in the market by the end of 2031, followed by Europe, North America, and LAMEA.
The innovative drug developments due to the increasing patent expiration of critical pharmaceuticals, as well as the growing outsourcing of formulation development services by most biotechnology and pharmaceutical companies the upcoming trends in the market.
The leading application of formulation development outsourcing market includes its use for development of vaccines.
North America is the largest regional market for formulation development outsourcing.
The formulation development outsourcing industry is estimated to reach $6,518.8 million by 2031
Charles River Laboratories, Syngene International Limited, and Catalent, Inc. are the top companies in formulation development outsourcing to hold the highest market share.
Loading Table Of Content...



