Lipid Nanoparticles Market Research, 2033
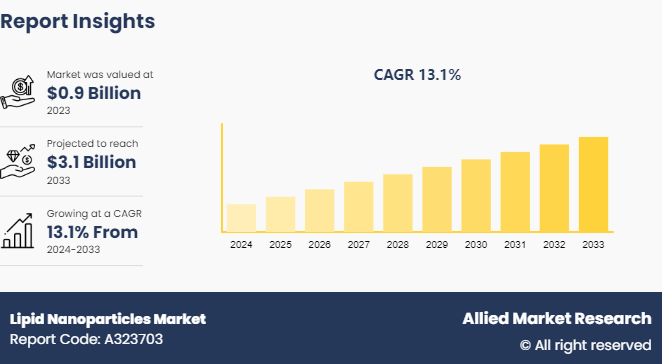
The global lipid nanoparticles market size was valued at $0.9 billion in 2023, and is projected to reach $3.1 billion by 2033, growing at a CAGR of 13.1% from 2024 to 2033. Increase in adoption of mRNA-based therapeutics, rise in investment in biotechnology & pharmaceutical research, surge in demand for targeted and controlled drug delivery systems, and technological advancements in nanomedicine are key factors driving the growth of the lipid nanoparticles market.
Market Introduction and Definition
The lipid nanoparticles (LNPs) market encompasses the design, formulation, and commercialization of lipid-based nanoscale carriers for delivering bioactive molecules such as drugs, vaccines, or genetic material. Lipid nanoparticles improve stability, enable controlled release, and facilitate targeted delivery, enhancing the efficacy and safety of therapeutics. LNPs have gained significant attention due to their pivotal role in mRNA-based vaccines, gene therapies, oncology treatments, and regenerative medicine.
LNP development involves formulation of lipid mixtures, encapsulation of therapeutic payloads, and rigorous characterization to ensure particle size, stability, and bioavailability. Advanced manufacturing platforms, analytical testing, and quality control procedures are essential to maintain consistency and meet regulatory standards. LNP applications span infectious diseases, cancer therapeutics, gene editing, and personalized medicine.
Rising adoption of mRNA vaccines, expansion of biopharmaceutical R&D, and technological innovations in lipid chemistry and scalable manufacturing are driving market growth. Collaborations between biotech firms, pharmaceutical companies, and contract development organizations (CDMOs) are further accelerating product development and commercialization. Regulatory frameworks and quality standards for nanoparticle safety and clinical translation are also enhancing market reliability.
As global demand for advanced therapeutics continues to rise, the lipid nanoparticles market is projected to witness steady growth between 2024 and 2033, supported by increasing therapeutic adoption, technological innovation, and expanding healthcare initiatives worldwide.
Key Takeaways
- The lipid nanoparticles market share analysis spans 20 countries, with detailed segmentation of each country in terms of market value over the forecast period.
- Over 1,500 sources—including product literature, industry announcements, annual reports of leading lipid nanoparticles companies, as well as authoritative industry journals, trade association publications, and government websites—were reviewed to generate comprehensive and reliable market insights.
- The study combines high-quality data, expert opinions, and independent critical analysis to offer a well-rounded perspective of the global market. This research approach is designed to provide stakeholders with actionable intelligence, enabling informed decision-making and supporting the achievement of ambitious growth objectives.
Key Market Dynamics
The lipid nanoparticles market is influenced by the rising adoption of mRNA-based therapeutics and vaccines, growing investment in biotechnology and pharmaceutical R&D, and increasing demand for advanced drug delivery systems. Expanding global biopharmaceutical infrastructure, technological innovations in lipid formulation, and regulatory guidance on nanoparticle safety are significantly shaping market growth. Additionally, the surge in gene therapies, oncology treatments, and personalized medicine is driving the need for effective lipid-based delivery platforms.
Drivers
The lipid nanoparticles market is primarily driven by the rapid expansion of mRNA therapeutics and vaccine development. The success of lipid nanoparticle-based COVID-19 vaccines has highlighted the critical role of LNPs in delivering nucleic acids safely and efficiently, boosting demand for both clinical and commercial applications. Pharmaceutical and biotechnology companies increasingly rely on LNPs for gene therapies, cancer treatments, and regenerative medicine, where targeted delivery and controlled release are essential for therapeutic efficacy.
Technological innovation is another key growth driver. Advancements in lipid chemistry, high-throughput formulation techniques, and automated manufacturing platforms enable more consistent particle size, encapsulation efficiency, and stability. Moreover, growing global collaborations between contract development and manufacturing organizations (CDMOs), biotech firms, and research institutions are accelerating the commercialization of lipid nanoparticle therapeutics. Regulatory frameworks and quality standards for nanoparticle safety, characterization, and clinical translation further reinforce market expansion by ensuring reliability and compliance.
Restraints
Despite strong growth potential, the lipid nanoparticles market faces certain challenges. High production costs and complex formulation requirements can limit adoption, particularly for small-scale pharmaceutical developers. Scaling up manufacturing while maintaining batch-to-batch consistency remains a technical challenge. Additionally, variations in regulatory guidance across regions may slow product approval and commercialization timelines. Limited infrastructure and expertise in nanoparticle formulation in emerging markets can also restrict market penetration.
Opportunities
The lipid nanoparticles market presents significant opportunities driven by ongoing digital transformation and expanding biopharmaceutical infrastructure in emerging economies. Integration of advanced analytics, artificial intelligence, and predictive modeling into LNP design and manufacturing can improve efficiency and therapeutic outcomes. Rising investments in personalized medicine, gene editing, and oncology therapeutics are expected to create new demand for lipid-based delivery systems. Strategic partnerships between biotech companies, research institutions, and contract manufacturers can further enhance production capabilities and global standardization. Increasing focus on regulatory compliance, patient safety, and the development of scalable, cost-effective LNP platforms is projected to accelerate long-term market growth between 2024 and 2033.
Market Segmentation
The lipid nanoparticles market analysis is segmented into type, application, end user, and region. On the basis of type, the market is categorized into solid lipid nanoparticles, nanostructured lipid carriers, and others. On the basis of application, the market is bifurcated into therapeutics and research. As per end user, the market is classified into pharmaceutical & biotechnology companies, academic & research institutes, and others. By region, it is analyzed across North America, Europe, Asia-Pacific, and LAMEA.
The lipid nanoparticles market exhibits diverse growth across segments. By type, solid lipid nanoparticles and nanostructured lipid carriers dominate, with other types contributing steadily. By application, therapeutics lead, followed by research. By end user, pharmaceutical and biotechnology companies hold the largest share, followed by academic and research institutes.
Regional/Country Market Outlook
North America leads the lipid nanoparticles market, driven by a robust pharmaceutical sector, advanced healthcare infrastructure, and substantial investments in biotechnology research. The presence of key industry players and early adoption of innovative drug delivery systems, particularly for mRNA vaccines, further strengthens the market in this region.
In the Asia-Pacific region, countries such as China, Japan, and India are witnessing rapid market expansion. This growth is fueled by increasing investments in healthcare infrastructure, rising awareness of advanced drug delivery technologies, and the growth of pharmaceutical manufacturing capabilities.
According to the National Nanotechnology Initiative (NNI) Supplement to the President's 2023 Budget, significant funding from agencies including the Department of Health and Human Services (HHS) and the Biomedical Advanced Research and Development Authority (BARDA) supports the development, validation, and commercialization of lipid nanoparticle-based mRNA delivery technologies.
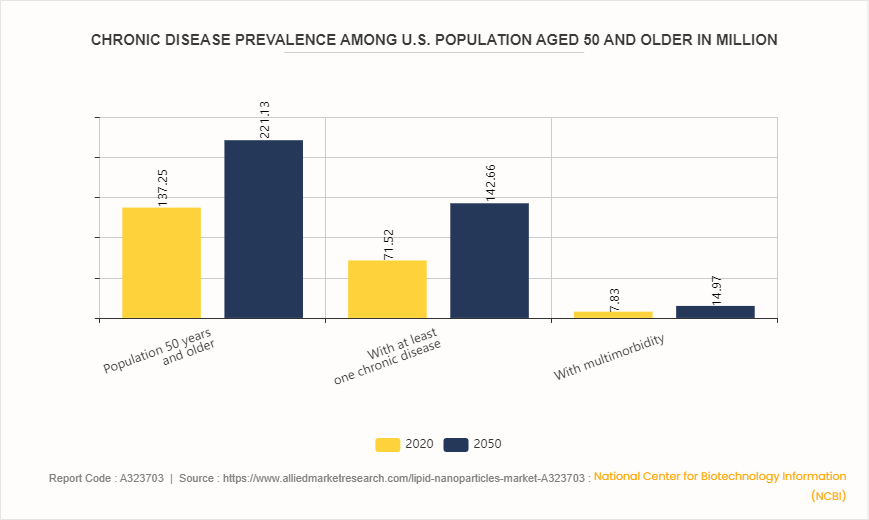
Chronic Disease Prevalence Among U.S. Population Aged 50 and Older
Projections from the National Center for Biotechnology Information (NCBI) indicate that the U.S. population aged 50 and above is expected to increase substantially, from 137.25 million in 2020 to 221.13 million by 2050. During the same period, the prevalence of chronic diseases within this demographic is projected to nearly double, with individuals living with at least one chronic condition rising from 71.52 million to 142.66 million. Additionally, the number of people experiencing multiple chronic conditions (multimorbidity) is expected to grow from 7.83 million to 14.96 million.
This rising burden of chronic diseases highlights the urgent need for advanced therapeutic approaches, such as lipid nanoparticle-based drug delivery systems, which enable targeted therapy and enhanced treatment effectiveness. Consequently, the lipid nanoparticles market is well-positioned for growth, driven by the increasing demand for innovative solutions to manage complex health conditions among the aging population.
Key End-Users Driving Growth in the Lipid Nanoparticles Market
The expansion of the lipid nanoparticles market is primarily fueled by pharmaceutical and biotechnology companies, academic and research institutes, and contract development and manufacturing organizations (CDMOs). Pharmaceutical and biotechnology firms hold the largest share, leveraging LNPs for mRNA vaccines, gene therapies, oncology drugs, and other advanced therapeutics. Academic and research institutions contribute significantly through preclinical studies, formulation development, and mechanistic research. CDMOs support large-scale production, ensuring consistent quality and regulatory compliance for commercial LNP products.
Major Trends Shaping the Lipid Nanoparticles Industry
A key trend in the lipid nanoparticles market is the increasing adoption of scalable, automated formulation and manufacturing platforms. High-throughput encapsulation techniques, microfluidics-based systems, and continuous production technologies are improving consistency, efficiency, and reproducibility. Another trend is the growing focus on targeted delivery and personalized medicine, with LNPs engineered to optimize pharmacokinetics, reduce toxicity, and enhance therapeutic efficacy. Global collaborations and licensing agreements are also accelerating R&D and commercialization of novel LNP-based therapeutics.
- In October 2022, the U.S. Food and Drug Administration (FDA) hosted a symposium to discuss the newly finalized guidance on drug products containing nanomaterials and its application in regulatory submissions.
- In the same year, the Australian government awarded a $3 million grant through the Cooperative Research Centres Projects Grants program to BioCina, a biopharmaceutical CDMO based in Adelaide, South Australia. The project, in collaboration with the University of Adelaide and Cytiva, focuses on advancing the design and manufacturing of lipid nanoparticles.
Role of Artificial Intelligence in the Lipid Nanoparticles Market
Artificial intelligence (AI) is increasingly applied in LNP development to optimize formulation design, predict stability, and enhance delivery efficiency. Machine learning models can analyze large datasets to identify optimal lipid compositions, particle sizes, and encapsulation strategies. AI-driven predictive modeling accelerates preclinical testing and reduces trial-and-error experimentation, while automated quality monitoring ensures consistent batch performance during manufacturing.
Technological Advancements Strengthening the Lipid Nanoparticles Market
Technological innovation in the lipid nanoparticles market focuses on advanced formulation methods, characterization tools, and integrated digital platforms. Microfluidics, high-pressure homogenization, and automated encapsulation systems enable precise control over particle size and payload delivery. Analytical technologies such as dynamic light scattering, electron microscopy, and HPLC-based assays ensure product quality. Integration with digital manufacturing platforms and LIMS allows real-time process monitoring, data traceability, and regulatory compliance, enhancing overall efficiency and reliability of LNP production.
Industry Trends
- The Food Drug & Administration (FDA) held a symposium in October 2022 to discuss the recently finalized guidance on drug products containing nanomaterials and how it can be implemented in regulatory filings.
- In 2022, the Australian government announced a $3 million grant through the Cooperative Research Centres Projects Grants scheme to BioCina, a biopharmaceutical contract development and manufacturing organization (CDMO) based in Adelaide, South Australia. BioCina’s partners in the project are the University of Adelaide and Cytiva. Researchers at the university will address the design and manufacture of lipid nanoparticles.
Competitive Landscape
The lipid nanoparticles market comprises a mix of global biopharmaceutical companies, specialized contract development and manufacturing organizations (CDMOs), and research-focused technology providers. Major players in this market include ABP Biosciences, LLC., Merck KGaA, Cytiva, CD Bioparticles, CordenPharma, Precigenome LLC., Beam Therapeutics, Acuitas Therapeutics, GENEVANT SCIENCES CORPORATION, and Helix Biotech, Inc. Other notable participants include Arbutus Biopharma, Croda International Plc, and regional innovators.
These organizations provide lipid nanoparticle formulation, encapsulation technologies, analytical characterization, and scalable manufacturing solutions for therapeutics, vaccines, and gene therapies. Market participants focus on technological innovation, strategic collaborations, clinical pipeline expansion, and global manufacturing outreach to strengthen their presence and meet growing demand for advanced drug delivery systems.
Recent Key Strategies and Developments in Lipid Nanoparticles Industry
- In February 2024, CordenPharma International introduced new Lipid NanoParticle (LNP) Starter Kits designed to support effective mRNA formulation for the development of mRNA-based therapeutics, including vaccines and gene therapies. The kits provide researchers and developers with essential components to create and optimize LNPs for efficient mRNA delivery.
- In July 2023, Cytiva, a global life sciences company, launched the NanoAssemblr commercial formulation system, specifically engineered for clinical and commercial manufacturing of lipid nanoparticle medicines.
- In August 2022, Merck partnered with Orna Therapeutics, a biotechnology firm pioneering engineered circular RNA therapies, to collaborate on the discovery, development, and commercialization of multiple programs, including vaccines and therapeutics targeting infectious diseases and oncology.
- In January 2022, Pfizer Inc. and Acuitas Therapeutics entered a Development and Option agreement, granting Pfizer a non-exclusive option to license Acuitas’ LNP technology for up to 10 targets for vaccine or therapeutic development, supporting the advancement of mRNA-based therapeutics.
Key Sources Referred
- National Center for Biotechnology and Information (NCBI)
- Centers for Medicare & Medicaid Services (CMS)
- National Health Service (NHS)
- Australian Government Department of Health and Aged Care
- Government of Canada's Health and Wellness
- Ministry of Health and Family Welfare (MoHFW)
- National Health Mission (NHM)
- Centers for Disease Control and Prevention (CDC)
- Food and Drug Administration (FDA)
- National Institutes of Health (NIH)
- World Health Organization (WHO)
Key Benefits for Stakeholders
- This report offers a quantitative assessment of market segments, trends, forecasts, and dynamics in the lipid nanoparticles market from 2023 to 2035, identifying key opportunities for growth.
- The research provides insights into major market drivers, restraints, and opportunities shaping industry development.
- Porter’s Five Forces analysis evaluates the bargaining power of buyers and suppliers, helping stakeholders make strategic, profit-oriented decisions and optimize their supplier-buyer networks.
- Detailed segmentation analysis highlights prevailing opportunities across product types, applications, end users, and regions.
- Leading countries within each region are mapped based on their revenue contribution to the global lipid nanoparticles market.
- Market player positioning supports benchmarking and offers a clear view of the competitive landscape.
- The report also covers regional and global market size, key industry participants, market segments, application areas, and growth strategies, providing a comprehensive overview for informed business planning and investment decisions.
Lipid Nanoparticles Market Report Highlights
| Aspects | Details |
| Market Size By 2033 | USD 3.1 Billion |
| Growth Rate | CAGR of 13.1% |
| Forecast period | 2024 - 2033 |
| Report Pages | 220 |
| By Type |
|
| By Application |
|
| By End User |
|
| By Region |
|
| Key Market Players | Cytiva, CD Bioparticles, CordenPharma, ABP Biosciences, LLC., Acuitas Therapeutics., Precigenome LLC., Helix Biotech, Inc., Merck KGaA, Beam Therapeutics, Genevant Sciences Corporation |
The total market value of lipid nanoparticles market is $0.9 billion in 2023.
The market value of lipid nanoparticles market in 2033 is $3.1 billion.
The forecast period for lipid nanoparticles market is 2024 to 2033.
The base year is 2023 in lipid nanoparticles market.
Lipid Nanoparticles are tiny, spherical vesicles made from lipids (fats) that are used as drug delivery systems. They can encapsulate active pharmaceutical ingredients (APIs), such as mRNA, proteins, or small molecules, protecting them from degradation and ensuring efficient delivery to target cells.
The growth of the market is driven by the increasing demand for novel drug delivery systems, advancements in nanotechnology, the rise in mRNA-based vaccines and the growing application of LNPs in cancer therapy, gene therapy, and other chronic diseases.
Loading Table Of Content...



