Newborn Screening Instruments Market Research, 2033
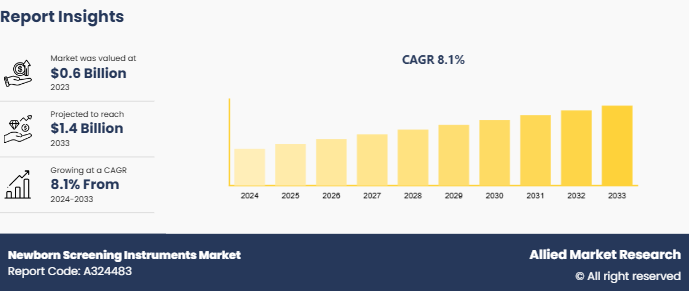
The global newborn screening instruments market size was valued at $0.6 billion in 2023, and is projected to reach $1.4 billion by 2033, growing at a CAGR of 8.1% from 2024 to 2033. The technological advancements in the screening devices such as improved accuracy, and integration with electronic health records, enhance the efficiency and reliability of the instruments and thereby drive the newborn screening instruments market growth.
Market Introduction and Definition
Newborn screening instruments are specialized medical devices used to detect congenital disorders, metabolic diseases, and genetic conditions in newborns shortly after birth. These instruments play a critical role in early diagnosis, enabling timely interventions that help prevent severe health complications, long-term disabilities, or death.
Key equipment includes mass spectrometers, pulse oximeters, hearing screening devices, and enzymatic assay systems. These tools analyze blood samples, oxygen levels, or auditory responses to identify potential abnormalities in newborns.
Newborn screening instruments are vital components of public health programs and are often supported by government mandates. They contribute to improved healthcare outcomes by enabling the early detection and treatment of conditions such as phenylketonuria (PKU), hypothyroidism, and cystic fibrosis.
Newborn screening instruments are widely used in hospitals, diagnostic laboratories, and neonatal care units to identify congenital, metabolic, genetic, and endocrine disorders in newborns. These instruments analyze blood samples, oxygen saturation, and auditory responses to detect conditions such as phenylketonuria (PKU), congenital hypothyroidism, cystic fibrosis, and critical congenital heart defects. They also support early hearing detection programs using specialized screening devices. By enabling rapid and accurate diagnosis shortly after birth, these instruments help healthcare providers initiate timely treatment and strategies to manage defects. As a result, newborn screening instruments play a vital role in reducing infant mortality, preventing long-term disabilities, and improving overall child health outcomes.
Key Takeaways
- The newborn screening instruments market share study covers 20 countries. The research includes a segment analysis of each country in terms of value ($Billion) for the projected newborn screening instruments market forecast period 2024-2033.
- More than 1, 500 product type literatures, industry releases, annual reports, and other such documents of major newborn screening instruments industry participants along with authentic industry journals, trade associations' releases, and government websites have been reviewed for generating high-value industry insights.
- The study integrated high-quality data, professional opinions and analysis, and critical independent perspectives. The research approach is intended to provide a balanced view of global markets and to assist stakeholders in making educated decisions in order to achieve their most ambitious growth objectives.
Key Market Dynamics
The growth of the newborn screening instruments market is influenced by several key drivers and restraints. A major factor driving market expansion is the increasing incidence of congenital disorders. Newborns are being diagnosed more frequently with conditions such as metabolic disorders, hearing impairment, and congenital hypothyroidism, which require early detection for effective management. Timely screening helps prevent long-term complications and improves quality of life, thereby increasing the demand for advanced screening instruments. In addition, growing awareness among parents and healthcare professionals regarding the importance of early diagnosis further supports market growth.
However, the high initial cost of newborn screening instruments remains a significant restraint. Advanced technologies, such as tandem mass spectrometry and genetic testing platforms, require substantial investment for equipment procurement, installation, and maintenance. Moreover, operational expenses related to training healthcare professionals and maintaining the necessary infrastructure can limit adoption, particularly in low-resource settings and developing countries.
Government initiatives and regulatory mandates play a crucial role in supporting market development. Many countries have implemented mandatory newborn screening programs backed by funding and policy frameworks, ensuring wider adoption of screening practices and improving access to early diagnosis. In addition, ongoing technological advancements—such as improved diagnostic accuracy, faster turnaround times, and integration with electronic health records—are enhancing the efficiency and reliability of screening instruments. These improvements make screening processes more accessible and user-friendly, encouraging healthcare providers to adopt advanced systems.
Despite the challenges associated with high costs, the rising prevalence of congenital disorders, supportive government policies, and continuous technological advancements present strong growth opportunities for the newborn screening instruments market. Addressing cost barriers will remain essential to ensure broader adoption of these critical, life-saving technologies.
Why Is Demand Increasing in This Market?
Demand for newborn screening instruments is increasing due to the rising prevalence of congenital, metabolic, and genetic disorders, which require early detection for effective treatment and improved long-term outcomes. Growing awareness among parents, healthcare providers, and policymakers about the benefits of early diagnosis is driving adoption, particularly in regions with well-established public health programs.
Technological advancements, such as rapid whole genome sequencing, tandem mass spectrometry, and AI-powered diagnostic tools, have expanded the scope and accuracy of screening, making these instruments more efficient and reliable. Additionally, government initiatives and mandates supporting mandatory newborn screening programs in countries like the U.S., European nations, China, and India are boosting market growth.
The combination of increasing birth rates in emerging markets, rising healthcare expenditure, and the focus on preventive care and early intervention is fueling the demand for advanced newborn screening instruments globally.
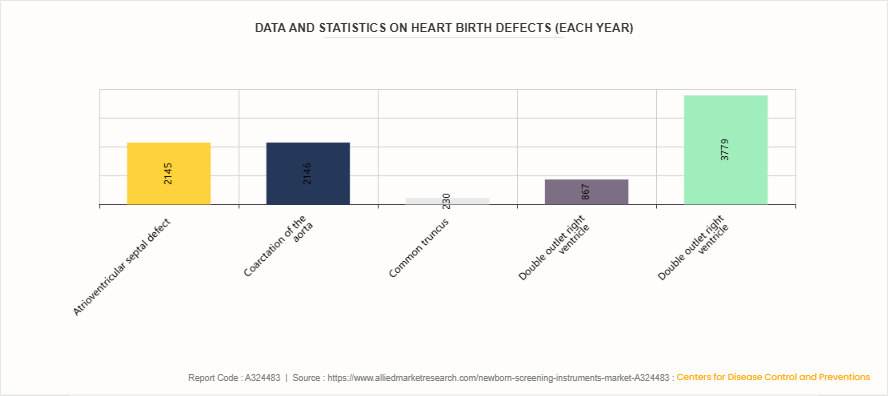
Data and Statistics on Heart Birth Defects (Each year)
According to data from the Centers for Disease Control and Prevention (CDC), several congenital heart defects affect thousands of newborns each year. Among these, atrioventricular septal defect and coarctation of the aorta are among the most common, affecting approximately 2,145 and 2,146 babies annually, respectively. Other conditions, such as common truncus, double outlet right ventricle, and pulmonary valve atresia and stenosis, impact around 230, 867, and 3,779 infants each year.
These figures highlight the critical importance of newborn screening instruments for early diagnosis and timely medical intervention. Early detection through screening programs plays a vital role in identifying congenital heart defects soon after birth, enabling prompt treatment and significantly improving health outcomes for affected infants.
Market Segmentation
The newborn screening instruments industry is segmented into product type, application, end user, and region. On the basis of the product type, the market is categorized into mass spectrometers, pulse oximeters, hearing screening devices, and enzyme analyzers. As per application, the market is segmented into tandem mass spectrometry, DNA-based assays, immunoassays, and others. On the basis of end user, the market is divided into hospitals, diagnostic centers, and others. Region wise, it is analyzed across North America, Europe, Asia-Pacific, and LAMEA.
Regional/Country Market Outlook
The regional and country outlook for the newborn screening instruments market highlights significant growth driven by advancements in healthcare infrastructure and supportive government initiatives. Developed regions such as North America and Europe dominate the market due to well-established newborn screening programs, continuous technological innovation, and high awareness regarding early disease detection.
In contrast, emerging economies in the Asia-Pacific and Latin America regions are experiencing rapid market growth, supported by increasing healthcare investments and improved access to screening services. Countries such as China, India, and Brazil offer lucrative opportunities due to their high birth rates and expanding neonatal care infrastructure. However, disparities in healthcare access, limited resources, and variations in the implementation of screening programs continue to pose challenges in certain regions.
Which Segment Holds the Largest Market Share and Why?
The mass spectrometry segment holds the largest market share in the newborn screening instruments market, primarily due to its widespread use, accuracy, and ability to simultaneously detect multiple metabolic and genetic disorders from a single sample. Tandem mass spectrometry (MS/MS), in particular, has become the standard in newborn screening programs globally, enabling rapid, reliable, and high-throughput analysis of amino acids, acylcarnitines, and other biomarkers. This capability allows early identification of conditions such as phenylketonuria (PKU), congenital adrenal hyperplasia, and other inborn errors of metabolism, which are critical for preventing severe complications.
Additionally, government mandates and public health initiatives in developed regions like North America and Europe have accelerated the adoption of mass spectrometry instruments in hospitals and diagnostic laboratories. The technology’s proven efficiency, low sample volume requirement, and ability to integrate with laboratory information systems further contribute to its dominance. As awareness of early diagnosis and preventive healthcare continues to rise, the mass spectrometry segment remains the preferred choice for large-scale, reliable newborn screening programs, reinforcing its leading market share.
Industry Trends
- In June 2022, Rady Children’s Institute for Genomic Medicine (RCIGM) announced a new program to advance and evaluate the scalability of a diagnostic and precision medicine guidance tool called BeginNGS. The tool is designed to screen newborns for approximately 400 genetic diseases with known treatment options using rapid whole genome sequencing (rWGS).
- BeginNGS was developed through a research collaboration with Alexion Pharmaceuticals, part of AstraZeneca’s rare disease group, along with Fabric Genomics, Genomenon, Illumina, and TileDB. The platform uses rWGS to diagnose genetic conditions and identify potential treatment options before symptoms appear. This approach represents a significant advancement over current pediatric applications of rWGS, which primarily focus on diagnosing children who are already critically ill.
What Makes the U.S./Europe/Asia-Pacific a Key Market for Industry Players?
The U.S., Europe, and the Asia-Pacific region represent key markets for newborn screening instruments due to their strong healthcare systems, government-supported screening programs, and growing demand for early disease detection technologies. In the U.S., national initiatives supported by organizations such as the Centers for Disease Control and Prevention and the Health Resources and Services Administration promote comprehensive newborn screening programs across states. Advanced healthcare infrastructure, high healthcare expenditure, and rapid adoption of innovative technologies such as genomic sequencing and tandem mass spectrometry further strengthen the market in the country.
Europe also plays a significant role due to well-established public healthcare systems and mandatory newborn screening policies in many countries. Regional collaboration through organizations such as the European Commission supports research, funding, and the harmonization of screening standards, driving the adoption of advanced screening technologies.
Meanwhile, the Asia-Pacific region is emerging as a high-growth market due to large birth populations, improving healthcare infrastructure, and increasing government investments in maternal and child healthcare. Countries such as China and India are expanding neonatal screening programs and healthcare facilities, creating strong demand for modern screening instruments and offering substantial opportunities for industry players.
What Technological Innovations Are Creating New Growth Opportunities?
Technological innovations are creating significant growth opportunities in the newborn screening instruments market by improving the accuracy, speed, and scope of early disease detection. One of the most impactful advancements is the adoption of next-generation sequencing (NGS) and rapid whole genome sequencing (rWGS), which enable healthcare providers to screen newborns for hundreds of genetic disorders simultaneously. For instance, initiatives by the Rady Children’s Institute for Genomic Medicine are advancing genomic-based screening programs that can detect rare genetic diseases even before symptoms appear, allowing earlier and more targeted interventions.
Another major innovation is the integration of artificial intelligence (AI) and data analytics in screening platforms. AI-powered algorithms help analyze large volumes of genetic and biochemical data, improving diagnostic accuracy and reducing false-positive results. In addition, advances in tandem mass spectrometry technology are enabling laboratories to screen multiple metabolic disorders from a single blood sample with greater efficiency.
Portable and noninvasive monitoring devices are also expanding screening capabilities beyond hospitals. Companies such as Masimo are developing smart baby monitoring systems that allow continuous health tracking at home. Together, these innovations are enhancing early detection, expanding screening coverage, and supporting the global adoption of advanced newborn screening technologies
What Is the Long-Term Outlook for the Market?
The long-term outlook for the newborn screening instruments market is highly positive, driven by growing awareness of early disease detection, increasing prevalence of congenital and genetic disorders, and ongoing technological advancements. Continuous innovations, including rapid whole genome sequencing, AI-enabled diagnostics, and portable monitoring devices, are expanding screening capabilities and improving accuracy. Supportive government policies, mandatory screening programs, and rising healthcare investments in both developed and emerging markets are expected to sustain market growth. Over the next decade, the market is expected to witness broader adoption, particularly in Asia-Pacific and Latin America, as healthcare infrastructure improves and early intervention becomes a global public health priority.
Competitive Landscape
The major players operating in the Newborn Screening Instruments Market Size include Natus Medical Incorporated, Trivitron Healthcare, Medtronic Inc., Bio-Rad Laboratories Inc., PerkinElmer, GE Healthcare, Masimo Corporation, Waters Corporation, Thermo Fisher Scientific, and Hill-Rom Holdings Inc.
Recent Key Strategies and Developments
- In August 2023, Masimo, a global leader in noninvasive monitoring technologies, announced the full U.S. market launch of the Stork smart home baby monitoring system. With the introduction of Stork, parents can now access technology originally developed for monitoring critically ill patients in hospitals. The system is designed with a simple and user-friendly interface, allowing parents to conveniently monitor their baby’s health through an easy-to-use mobile application on their smartphone.
Key Benefits for Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the newborn screening instruments market analysis from 2024 to 2033 to identify the prevailing newborn screening instruments market opportunity.
- The market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the newborn screening instruments market? segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global newborn screening instruments market trends, key players, market segments, application areas, and market growth strategies.
Newborn Screening Instruments Market , by Product Type Report Highlights
| Aspects | Details |
| Market Size By 2033 | USD 1.4 Billion |
| Growth Rate | CAGR of 8.1% |
| Forecast period | 2024 - 2033 |
| Report Pages | 163 |
| By Product Type |
|
| By Application |
|
| By End User |
|
| By Region |
|
| Key Market Players | Waters Corporation, Hill-Rom Holdings Inc., Bio-Rad Laboratories Inc., PerkinElmer, Medtronic Inc., trivitron healthcare pvt. ltd., Thermo Fisher Scientific Inc, GE Healthcare, Masimo Corporation, Natus Medical Incorporated. |
The total market value of Newborn Screening Instruments Market was $ 0.6 billion in 2023.
The market value of Newborn Screening Instruments Market is projected to reach $ 1.4 billion by 2033.
The forecast period for Newborn Screening Instruments Market is 2024 to 2033
The base year is 2023 in Newborn Screening Instruments Market.
Newborn screening instruments are specialized medical devices designed to detect congenital disorders, metabolic diseases, and genetic conditions in newborns shortly after birth. These instruments play a crucial role in early diagnosis, enabling timely interventions that can prevent severe health complications, disabilities, or death.
Loading Table Of Content...



