Patented Drugs Market Research, 2032
The global patented drugs market size was valued at $814.4 billion in 2022 and is projected to reach $1,456.5 billion by 2032, exhibiting a CAGR of 6.0% from 2023 to 2032. Patented drugs are pharmaceutical medications that have obtained legal protection and exclusivity by government authorities like the United States Patent and Trademark Office (USPTO), Intellectual Property Office, European Patent Office (EPO), and others. Patents provide exclusive rights to the patent holder for manufacturing, using, and selling the drug for a specified period, usually 20 years from the filing date. This protection prevents other companies from producing or marketing the same drug without the permission of the inventor.
Patented drugs represent the outcomes of substantial research and development endeavors, and patents serve to safeguard and incentivize innovation in the pharmaceutical industry. By granting exclusive rights, patents encourage investment in drug development and allow companies to recoup their investments while fostering competition once the patent protection expires.

Market Dynamics
The growth of patented drugs industry is driven by increase in R&D activities for novel medication development, rise in prevalence of chronic diseases, such as cardiovascular diseases, diabetes, cancer, and respiratory disorders, and increase in approvals of novel drugs. Through extensive research, scientists can identify potential drug targets, and conduct preclinical and clinical trials to test the safety and efficacy of new drugs. Such innovation leads to the creation of new patented drugs, which address unmet medical needs, improve treatment outcomes, and enhance patient care.
In addition, pharmaceutical companies invest in post-approval R&D activities to expand the therapeutic applications of their patented drugs and develop new formulations or explore combination therapies. These strategies aim to enhance the clinical benefits of existing drugs, extend their patent protection, and further drives the market growth. Furthermore, many pharmaceutical companies invest heavily in R&D, which is projected to boost the market growth. R&D investment allows pharmaceutical companies to expand and diversify their drug pipelines. This expanded pipeline increases the chances of successful drug development and market entry of new patented drugs. For instance, Biogen invested $2,231.1 million in R&D of new drugs.
In addition, increase in product approvals by regulatory authorities also drives the growth of the patented drugs market. Approved drugs are marketed and sold to healthcare providers, which expand their accessibility to patients. This market expansion leads to increased revenue generation which, in turn, supports the growth of the patented drugs market.
For instance, in December 2022, U.S. Food and Drug Administration (FDA), approved Sunlenca tablets developed by Gilead Sciences, Inc. for the treatment of HIV infections. Sunlenca is patented by U.S. Patent and Trademark Office (USPTO) in September 2018. Similarly, in October 2022, U.S. Food and Drug Administration (FDA), approved Imjudo injection developed by AstraZeneca for the treatment of hepatocellular carcinoma. Therefore, such product approvals are expected to drive the growth of the patented drugs market during the forecast period.
The growth of the patented drugs market is expected to be driven by increase in collaborations between pharmaceutical companies for the manufacturing and commercialization of patented drugs. This, in turn, contributes toward the growth of the patented drugs market. For instance, On March 14, 2022, Biogen, a global biopharmaceutical company entered into a collaboration agreement with Eisai for ADUHELM. Under this agreement, Biogen has complete rights on manufacturing and commercialization of ADUHELM. Furthermore, the pharmaceutical industry in emerging economies is developing at a significant rate, owing to a rise in demand for enhanced healthcare services, and an increase in prevalence of chronic diseases.
However, high cost of patented drugs and limited patent period restrains the growth of the patented drugs market. High costs of patented drugs may create barriers that restrict patient access to these innovative therapies, for individuals with low financial resources. Thus, limited access to costly patented drugs hampers the market growth. Furthermore, development and production of advanced drug requires substantial investments in research, technology, and infrastructure. This led to high development and production costs, which is projected to limit the entry of new pharmaceutical companies thereby restraining the market growth.
On the other hand, strong intellectual property rights and favorable patent filing process is expected to create new opportunities for the growth of patented drugs industry.
The outbreak of COVID-19 has disrupted workflows in the healthcare sector around the world. The disease has forced a number of industries to shut their doors temporarily, including several sub-domains of health care. The global patented drugs market was moderately impacted during the pandemic due to disruption in supply of pharmaceutical drugs and delay in R&D activities.
On the other hand, patent filing is increased during the pandemic for the novel Corona Virus vaccines. Furthermore, rise in demand for patented drugs and increase in prevalence of chronic diseases are expected to drive the market growth during the forecast period.
Segmental Overview
The patented drugs market is segmented on the basis of dosage form, indication, distribution channel, and region. On the basis of dosage form, the market is classified into solid drug product, liquid drug product, and others. On the basis of indication, the market is categorized into cardiovascular diseases, cancer, autoimmune diseases, and others.On the basis of distribution channel, the market is divided into hospital pharmacies, drug stores & retail pharmacies, and online pharmacies.
Region wise, the market is analyzed across North America (the U.S., Canada, and Mexico), Europe (Germany, France, the UK, Italy, Spain, and rest of Europe), Asia-Pacific (China, Japan, Australia, India, South Korea, and rest of Asia-Pacific), and LAMEA (Brazil, South Africa, Saudi Arabia, and rest of LAMEA).
By Dosage Form
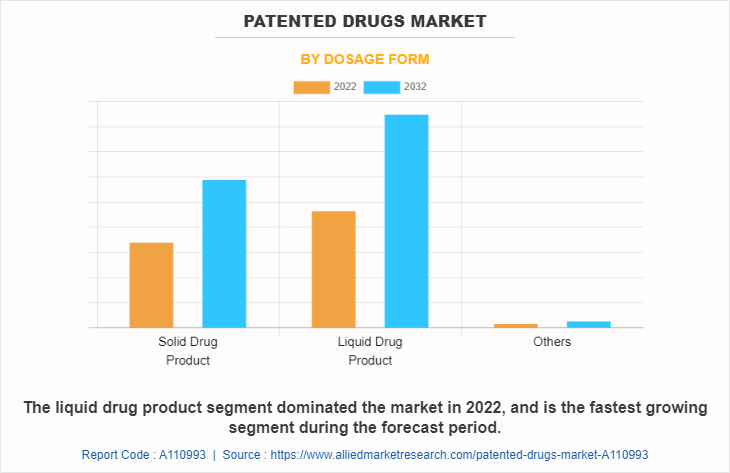
The patented drugs market is segmented into solid drug product, liquid drug product, and others. The liquid drug product segment accounted for the largest patented drugs market share in 2022, and is expected to remain dominant throughout the forecast period as liquid drugs are formulated to exhibit faster absorption rates and quicker onset of action compared to solid dosage forms.
By Indication
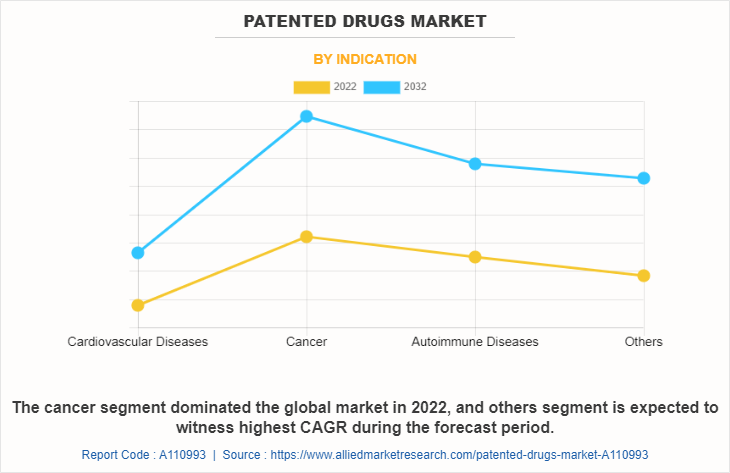
The patented drugs market is segregated into cardiovascular diseases, cancer, autoimmune diseases, and others. The cancer segment accounted for largest patented drugs market share in 2022 and is anticipated to continue this trend during the forecast period. This is attributed to wide availability of patented drugs for the treatment of cancer. On the other hand others segment is expected to register fastest CAGR growth during patented drugs market forecast period, owing to surge in research and development for rare diseases.
By Distribution Channel
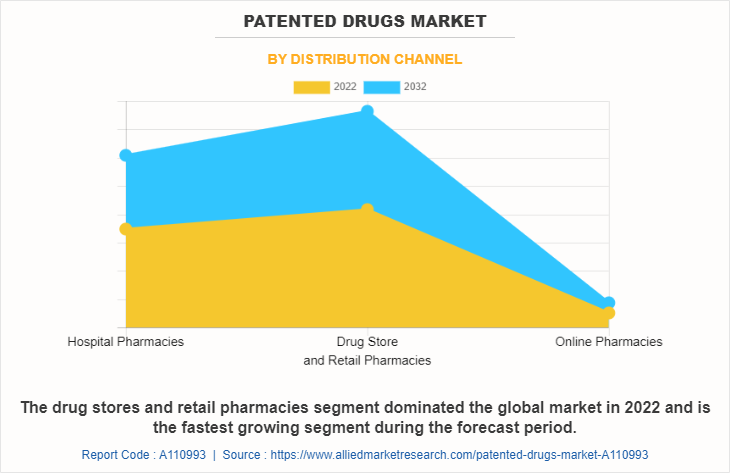
The patented drugs market is segmented into hospital pharmacies, drug stores & retail pharmacies, and online pharmacies. The drug stores & retail pharmacies segment dominated the global market in 2022 and is anticipated to remain dominant during the forecast period. This is attributed to increase in hospitalization rate and surge in demand for patented drugs through retail pharmacies and drug stores.
By Region
The patented drugs market is analyzed across North America, Europe, Asia-Pacific, and LAMEA. North America accounted for a major share of the patented drugs market size in 2022 and is expected to maintain its dominance during the forecast period.
The presence of several major players, such as Pfizer Inc., Novartis AG, F. Hoffmann La-Roche Ltd., and Amgen Inc. and rise in prevalence of various chronic diseases in the region drives the growth of the market. For instance, according to National Diabetes Statistics Report 2020 provided by Centers for Disease Control and Prevention (CDC), estimated that about 34.2 million people (10.5%) U.S. population have diabetes. Increase in prevalence of diabetes leads to greater demand for novel medications which drives the market growth.
In addition, strong presence of various cancer research institutes such as American Association for Cancer Research (AACR), Mayo Clinic Cancer Center, and National Cancer Institute (NCI) and strong research infrastructure, with a large number of research funding agencies has led to the development of a robust research ecosystem that supports the growth of new R&D projects. Moreover, easy accessibility and availability of patented drugs boost the growth of the market.
Asia-Pacific offers profitable patented drugs market opportunity, thereby registering the fastest growth during the forecast period. This is attributable to the increase in demand for prescription medications for the treatment of cancer and various cardiovascular diseases. Asia-Pacific region has a large population, and there has been an observed increase in the prevalence of cancer in recent years due to ageing population, lifestyle changes, and environmental factors. This has led to an increase in the number of patients undergoing treatment, which in turn increases the demand for patented drugs.
For instance, in 2020, National Institute of Cancer Prevention and Research (NICPR), estimated that about 2.7 million people in India are living with cancer. In addition, growing adoption of patented drugs further propels the market growth. Asia-Pacific offers profitable opportunities for key players operating in the patented drugs market, thereby registering the fastest growth rate during the forecast period, owing to the rise in disposable income, as well as increase in healthcare expenditure. Moreover, development of pharmaceutical industry and increase in R&D activities boost the market growth in this region.

Competition Analysis
Competetion analysis and profiles of the major players in the patented drugs, such as AbbVie Inc., Amgen Inc., Pfizer Inc., Novartis AG, AstraZeneca plc, Bayer AG, Johnson & Johnson, F. Hoffmann-La Roche AG, Astellas Pharma Inc., and Biogen. Major players have adopted patent and patent extension as key developmental strategies to improve the product portfolio and gain strong foothold in the patented drugs market.
Recent Patent Grant in the Patented Drugs Market
In June 2022, Biogen, a leading global biotechnology company received a patent grant from the European Patent Office (EPO) to TECFIDERA for the treatment of relapsing forms of multiple sclerosis that expires in February 2028.
In February 2022, Novartis AG, one of the global leaders in pharmaceutical industry received patent grant from the European Patent Office (EPO) for Gilenya (fingolimod) 0.5mg daily dose.
Recent Patent Extension in the Patented Drugs Market
In February 2021, Pfizer Inc., a global pharmaceutical company announced that the U.S. Patent and Trademark Office (USPTO) issued a U.S. Patent Term Extension (PTE) certificate for IBRANCE.
Key Benefits For Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the patented drugs market analysis from 2022 to 2032 to identify the prevailing patented drugs market opportunities.
- The market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the patented drugs market segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global patented drugs market trends, key players, market segments, application areas, and market growth strategies.
Patented Drugs Market Report Highlights
| Aspects | Details |
| Market Size By 2032 | USD 1456.5 billion |
| Growth Rate | CAGR of 6% |
| Forecast period | 2022 - 2032 |
| Report Pages | 263 |
| By Distribution Channel |
|
| By Dosage Form |
|
| By Indication |
|
| By Region |
|
| Key Market Players | F. Hoffmann-La Roche AG, Johnson & Johnson, Biogen, Astellas Pharma Inc., AstraZeneca plc, Pfizer Inc., Novartis AG, AbbVie Inc., Amgen Inc., Bayer AG |
Analyst Review
Patented drugs are pharmaceutical products that have been granted exclusive rights through a patent. These patents protect the drugs from being manufactured, marketed, or sold by other pharmaceutical companies without permission. Increase in patent extension and patenting strategies drive the growth of the patented drugs market.
Furthermore, patent filing and patented extension strategies adopted by the key market players are expected to create opportunities for market growth. For instance, in February 2022, Novartis AG, one of the global leader in pharmaceutical industry, received patent grant from the European Patent Office (EPO) for Gilenya (fingolimod) 0.5mg daily dose. Furthermore, in February 2021, Pfizer Inc., a global pharmaceutical company, announced that the U.S. Patent and Trademark Office (USPTO) issued a U.S. Patent Term Extension (PTE) certificate for IBRANCE. Such patent grants and patent extension boost the growth of the patented drugs market.
A patent is a form of legal protection granted by a government authority to inventors for their novel drug product.
The major factor that fuels the growth of the patented drugs market are surge in investment in research and development of new drugs, increase in patent filing activity, and rise in disease burden.
Liquid drug product is the most influencing segment in patented drugs market which is attributed to availability of liquid drug product across globe and increase in product launches for liquid drug product.
Top companies such as, AbbVie Inc., AstraZeneca plc, Novartis AG, and F. Hoffmann-La Roche AG held a high market position in 2022. These key players held a high market position owing to the strong geographical foothold in different regions.
The total market value of patented drugs market is $814.5 billion in 2022.
The forecast period for patented drugs market is 2023 to 2032.
The market value of patented drugs market in 2032 is $1,456.4 billion.
The base year is 2022 in patented drugs market.
Loading Table Of Content...
Loading Research Methodology...



