Percutaneous Transluminal Coronary Angioplasty (PTCA) Balloon Catheters Market Research, 2031
The global percutaneous transluminal coronary angioplasty (ptca) balloon catheters market size was valued at $1.2 billion in 2021, and is projected to reach $1.8 billion by 2031, growing at a CAGR of 4.4% from 2022 to 2031. A minimally invasive treatment known as PTCA is used to unblock blocked or restricted coronary arteries caused due to coronary artery disease (CAD). PTCA balloon catheters allow blood to flow freely from the arteries to the heart muscles without the need of open-heart surgery. Smoking, high blood pressure, diabetes, and high cholesterol level in the blood all contribute to coronary heart disease. Chest pain, sweating, difficulty in breathing, dyspnea, and severe fatigue are some of the signs of coronary heart disease. Some of the primary factors driving the PTCA Balloon Catheters Market growth are geriatric population, technological improvements, and introduction of innovative products.
Market Dynamics
The drivers of percutaneous transluminal coronary angioplasty balloon catheters market include its simplicity to use, safety, and cost-effective. Guiding catheters are used to show when interventional devices are being delivered to the peripheral or coronary vascular systems. The fundamental benefit of PTCA is that it eliminates the immediate post-operative discomforts associated with coronary bypass surgery while lowering morbidity. The global PTCA balloon catheter market is being driven by increase in geriatric population and surge in the occurrence of cardiovascular disease. The geriatric population is expected to fuel the expansion of the PTCA balloon catheters market, owing to the risk of cardiovascular problems that increase with age.
High cholesterol levels in the blood, smoking, obesity, high blood pressure, and other variables linked with the risk of developing heart disease fuel the expansion of the PTCA balloon catheters market. However, as policies and standards are developed to ensure the safety of patients, the number of procedures will continue to rise. Revisions to national and international standards have been issued to assure the utmost safety of patients and healthcare workers, as well as to eliminate delays in chronic illness management care. Despite lower patient admissions in 2021, the PTCA balloon catheters market is expected to grow in the next years, due to a decrease in COVID-19 cases and a large proportion of the population suffering from chronic diseases.
The World Health Organization (WHO) on January 30, 2021 declared COVID-19 as pandemic. Coronavirus has impacted around 210 countries across the globe. State run administrations across the globe declared inescapable lockdowns as well as friendly distancing measures to forestall breakdown of the healthcare structure. States have additionally given constraints and preventions on undertakings and elective surgeries. These obstructions continue to influence the growth of different industries, and the quick effect of the episode changes starting with one industry then onto the following. The scattering, production, and store network have been impacted, because of the execution of lockdown across the globe.
The pandemic of COVID-19 had a negative impact on PTCA balloon catheters market growth. During the COVID-19 pandemic, there was a decline in the frequency of emergency and general visits linked to cardiovascular disorders, which influenced product acceptance to some extent. Several data reveal that the frequency of elective cardiac, neurology, and urology surgeries for the management of certain chronic diseases has decreased since the social distancing standards and new operating protocols at healthcare facilities were effective in early 2021. Hence, overall COVID had a negative impact on the market.
Segments Overview
The percutaneous transluminal coronary angioplasty (ptca) balloon catheters market is segmented into Product, Delivery Platform, Compliance and Balloon Material. The global PTCA balloon catheter market is segmented on the basis of product, , delivery platform, compliance, balloon material. On the basis of product , it is segmented into normal balloon catheter, drug eluting balloon catheter, cutting balloon catheter, and scoring balloon catheter. Depending on delivery platform, itis fragmented into over-the wire and rapid exchange/monorail balloon catheters. According to compliance, it is categorized into semi-compliant and non-compliant. As per balloon material, it is classified into polyolefin copolymer (POC), polyethylene (PE) and polyethylene terephthalate (PET) Region wise, the PTCA balloon catheters market is analyzed across North America, Europe, Asia-Pacific, and LAMEA.
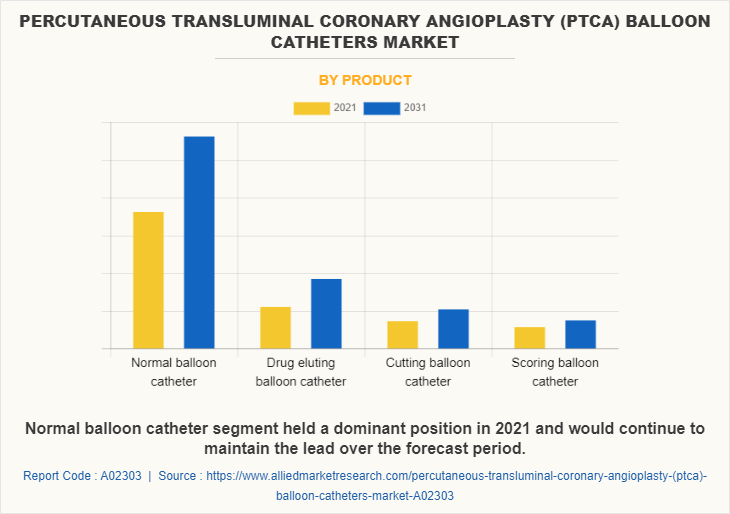
By Product
Based on product, in PTCA balloon catheters market trends normal balloon catheter is projected to record a 4.5% CAGR and reach $1125.56 million by the end of the PTCA Balloon Catheters Market forecast period and the growth in the drug eluting balloon catheter segment is readjusted to a revised 5.3% CAGR for the next 10-year period.
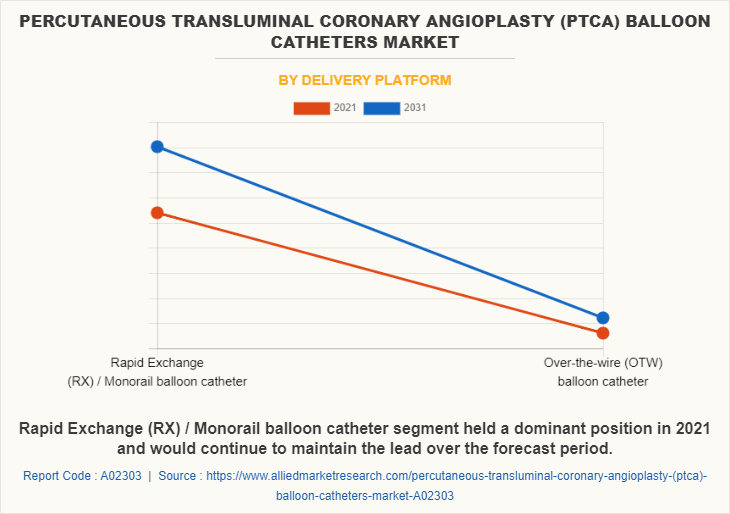
By Delivery Platform
On the basis of delivery platform, in PTCA balloon catheters market analysis Rapid exchange balloon catheters that have guidewire along a short section which helps in saving time has been reported as the largest segment with a revenue of 1,080.9 million in 2021. Whereas, over-the wire balloon catheters feature guidewire that tracks along the full length of catheter with a CAGR of 7.1% in PTCA balloon catheters market share during the forecast period.
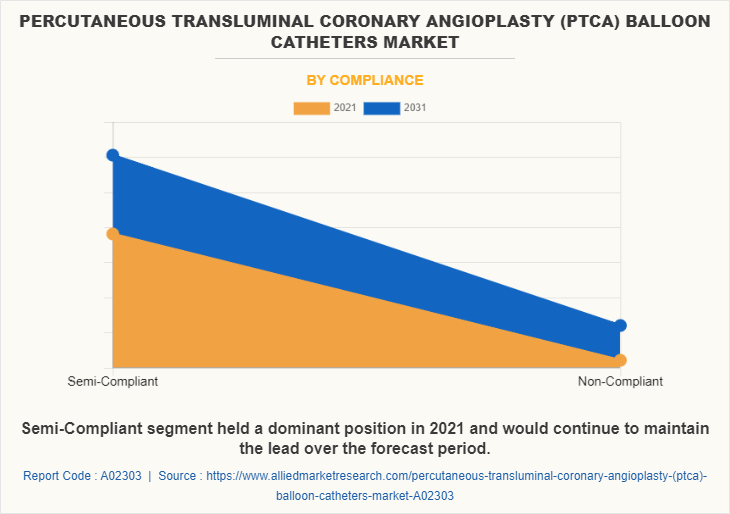
By Compliance Type
Based on compliance the PTCA balloon catheters market size is categorized into semi-compliant and non-compliant. The non –compliant balloons are made of polyester or nylon. They are used in applications where balloon needs to expand to a specific diameter and exert high pressure. The compliant balloons are made of polyurethane and silicone. They are able to stretch from 100% to 800% which makes them compatible to occlude the anatomy.
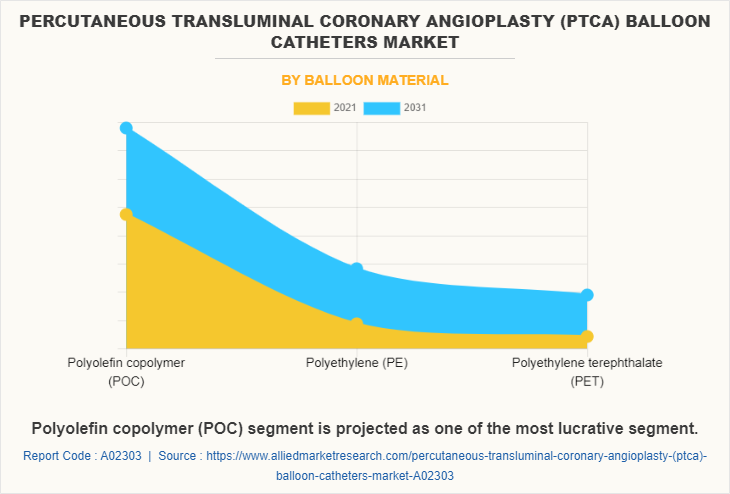
By Balloon Material
Based on balloon material, the PTCA balloon catheters industry is fragmented into polyolefin copolymer (POC), polyethylene (PE) and polyethylene terephthalate (PET). The Polyolefin copolymer (POC) segment dominated the market with a revenue of $672.35 million and is accounted to be the largest segment in 2021 and Polyethylene (PE) is accounted to be the fastest growing segment with a CAGR of 5.3% in the given forecast period.
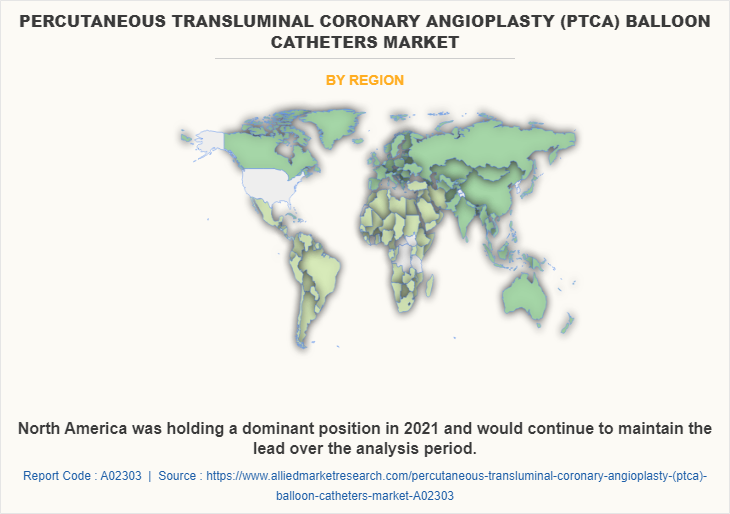
By Region
Region wise, North America has the largest market for PTCA balloon catheters. The PTCA balloon catheter market in North America is followed by Europe.
Due to rise in occurrences of coronary heart disease, rise in disposable income, increase in knowledge of heart diseases and associated treatments, and better healthcare infrastructure in Asia, the PTCA balloon catheter market is expected to grow rapidly in the Asia pacific in future.
The list of key players profiled in this report include Abbott Laboratories, B. Braun Melsungen AG, BIOTRONIK SE & Co. KG, Boston Scientific Corporation, C. R. Bard, Inc., Cardinal Health, Inc., Medtronic plc., MicroPort Scientific Corporation, Terumo Corporation, and THE SPECTRANETICS CORPORATION.
Key players in the value chain include Lepu Medical Technology (Beijing) Co., Ltd., Meril Life Sciences Pvt. Ltd., and COOK MEDICAL INC.
Key Benefits for Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the percutaneous transluminal coronary angioplasty (ptca) balloon catheters market analysis from 2021 to 2031 to identify the prevailing percutaneous transluminal coronary angioplasty (ptca) balloon catheters market opportunities.
- The market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the percutaneous transluminal coronary angioplasty (ptca) balloon catheters market segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global percutaneous transluminal coronary angioplasty (ptca) balloon catheters market trends, key players, market segments, application areas, and PTCA balloon catheters market opportunity growth strategies.
Percutaneous Transluminal Coronary Angioplasty (PTCA) Balloon Catheters Market Report Highlights
| Aspects | Details |
| By Product |
|
| By Delivery Platform |
|
| By Compliance |
|
| By Balloon Material |
|
| By Region |
|
| Key Market Players | KONINKLIJKE PHILLIPS, ABOTT LABORATORIES, B.BRAUN MELSUNGEN AG, MEDTRONIC PLC, BALTON SP ZOO, BOSTON SCIENTIFIC CORPORATION, BECTON, DICKINSON & COMPANY, ALVI MEDICA, IVASCULAR, RONSTIS |
Analyst Review
A minimally invasive treatment called percutaneous transluminal coronary angioplasty (PTCA) is performed to unblock blocked or constricted coronary arteries. PTCA balloons are inflated inside a clogged coronary artery, to open the blockage and allow easy blood flow. PTCA balloon catheters allow blood to flow freely from the arteries to the heart muscles without the need of open heart surgery.
Smoking, high blood pressure, diabetes, and high cholesterol level in the blood all contribute to coronary heart disease along with chest pain, sweating, difficulty in breathing, dyspnea, and severe fatigue are some of the signs of coronary heart disease. Some of the primary factors driving the PTCA Balloon Catheters Market growth of the PTCA balloon catheter market are an older population, technological improvements, and the introduction to innovative products.
One of the primary reasons driving market value is the expanding geriatric population, which is accompanied by an increase in the prevalence of aortic disease, venous thromboembolism (VTE), and peripheral arterial disease (PAD) due to age-related changes in vascular structure and function. The number of elderly individuals in Asia-Pacific is increasing at an unprecedented rate, and the region's ageing population has already reached critical mass.
In 2020, North America has the largest market for PTCA balloon catheters industry because of increased healthcare infrastructure, government initiatives, an increase in the rates of cardiac diseases, and rising healthcare expenditures in this region.
The top companies include Abbott Laboratories, B. Braun Melsungen AG, BIOTRONIK SE & Co. KG, Boston Scientific Corporation, C. R. Bard, Inc., Cardinal Health, Inc., Medtronic plc, MicroPort Scientific Corporation, Terumo Corporation, and THE SPECTRANETICS CORPORATION.
North America is the largest regional market in PTCA market.
The leading application of Percutaneous Transluminal Coronary Angioplasty (PTCA) Balloon Catheters Market includes peripheral vascular diseases and cardiovascular disorders.
The trends include increase in ageing population and changes in lifestyle leading to hypertension, obesity, and other risk factors that lead to vascular disorders is expected to propel the growth of the PTCA balloon catheters market
The global percutaneous transluminal coronary angioplasty (PTCA) balloon catheters market was estimated to be $1,2021.23 million in 2021 and is estimated to reach $1,848.76 million in 2031 with an expected CAGR of 4.4% from 2021 to 2031.
Loading Table Of Content...



