Pyrogen Testing Market Oerview
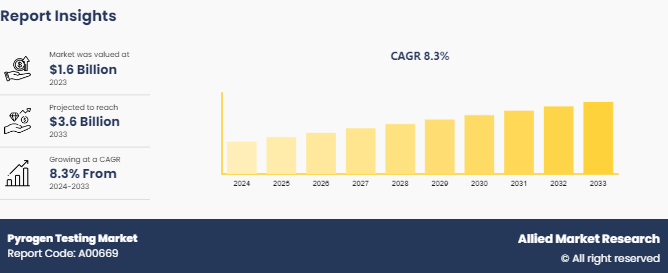
The global pyrogen testing market size was valued at $1.6 billion in 2023, and is projected to reach $3.6 billion by 2033, growing at a CAGR of 8.4% from 2025 to 2033. The growth of the pyrogen testing market is driven by increase in demand for pharmaceutical and biotechnology products, rise in investments in research and development, and surge in need for ensuring the safety of injectable drugs and medical devices. The developments in biopharmaceutical industry and rise in prevalence of chronic diseases also foster the need for pyrogen testing.
Market Size & Future Outlook
- 2023 Market Size: $1.6 Billion
- 2033 Projected Market Size: $3.5 Billion
- CAGR (2024-2033): 8.34%
- North America: Largest market in 2023
- Asia Pacific: Fastest growing market
What is Meant by Pyrogen Testing
Pyrogen testing is an important procedure across the healthcare industry. It is adopted to ensure the safety and efficacy of medical products and pharmaceuticals. Pyrogens are fever-inducing substances that can be present in injectable medications, medical devices, and other products. They are often microbial toxins, such as endotoxins produced by gram-negative bacteria, which can cause severe reactions if introduced into the human body. Pyrogen testing is used to detect and quantify contaminants. The major factors driving the growth of the pyrogen testing market are increase in pharmaceutical and biotechnology activities, stringent regulatory requirements for pharmaceutical and medical devices, and rise in awareness of the potential risks associated with pyrogens in medical products. In addition, rise in prevalence of chronic diseases and rise in drug development activities create a higher demand for pyrogen testing.
Key Takeaways
- The pyrogen testing market share study covers 20 countries. The research includes a segment analysis of each country in terms of value for the projected period.
- More than 1, 500 product literatures, industry releases, annual reports, and other such documents of major Pyrogen Testing industry participants along with authentic industry journals, trade associations' releases, and government websites have been reviewed for generating high-value industry insights.
- The study integrated high-quality data, professional opinions and analysis, and critical independent perspectives. The research approach is intended to provide a balanced view of global markets and to assist stakeholders in making educated decisions in order to achieve their most ambitious growth objectives
What Is the Current Market Size and How Is the Market Expected to Grow?
The steady growth of the pyrogen testing market is attributed to increase in need for pharmaceutical safety, biologics development, and regulatory compliance. Pyrogen testing helps ensure that pharmaceutical drugs, vaccines, biologics, medical devices, and injectable products are free from pyrogens, which are substances that can cause fever or severe immune reactions in humans. Regulatory agencies such as the FDA, EMA, and other global authorities require strict pyrogen testing before product approval. Further, rise in development of biologics, biosimilars, and advanced therapies also foster the growth of the market. These products are highly sensitive and require rigorous safety testing to prevent contamination.
Region-wise, North America holds the largest share due to presence of advanced pharmaceutical manufacturing and regulatory frameworks. Asia-Pacific is expected to witness the fastest growth due to developments in pharmaceutical industries in China and India.
Key Market Dynamics
According to the pyrogen testing market forecast analysis the key factors driving the growth of the market are regulatory requirements, growth in biopharmaceuticals industry, technological advancement, and surge in research and development. Regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) mandate stringent pyrogen testing protocols to ensure the safety and efficacy of biopharmaceutical products. These regulations require manufacturers to conduct pyrogen tests on injectable drugs and medical devices to detect bacterial endotoxins, which can cause severe adverse reactions in patients. According to the UK Government, the UK life sciences industry grew about 13% from 2021 to 2022. The growth of this industry makes way for advancements in pyrogen testing technologies. Thus, companies seek to meet regulatory standards and maintain product safety.
In addition, according to the pyrogen testing market Growth analysis, technological advancement and a surge in research and development (R&D) are pivotal drivers in the pyrogen testing market. As technology continues to evolve, the development of more sophisticated and precise testing methods enhances the ability to detect pyrogens with greater accuracy and speed. This includes advancements in techniques such as the Limulus Amebocyte Lysate (LAL) test and recombinant Factor C (rFC) assays, which offer improved sensitivity and reliability.
What Are the Key Factors Driving and Restraining Market Growth?
Expansion of the pharmaceutical and biotechnology industries is one of the major factors that foster the growth of the pyrogen testing market. There is an increase in need for safety testing with development of injectable drugs, vaccines, and biologics. Further, strict regulatory requirements stated by health authorities also boost the demand for pyrogen testing before approval of drugs and medical devices. There is an increase in shift toward in vitro testing methods owing to their efficiency. Moreover, these methods also help reduce reliance on animal-based testing.
However, one key challenge is the high cost of advanced testing equipment and reagents in developing countries. Also, the complexity of testing procedures, which requires trained professionals and strict quality control systems deters the growth of the market. Regulatory variations across regions decrease adoption of pyrogen testing in emerging markets.
Pyrogen Test by Test Type
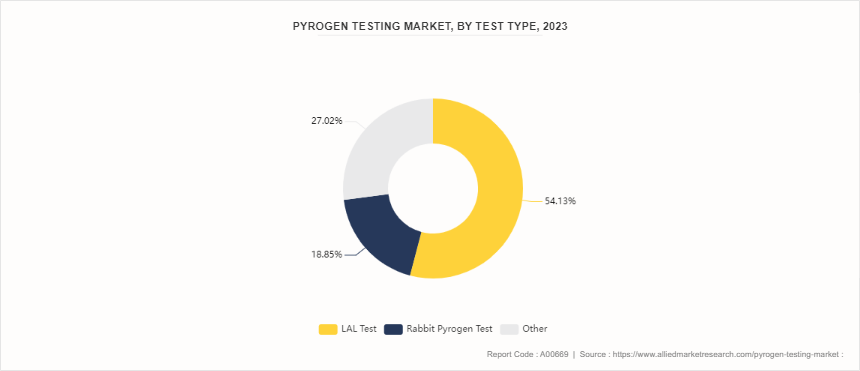
Pyrogen testing is a set of procedures and assays designed to detect the presence of pyrogens in pharmaceuticals, medical devices, and other healthcare products. Pyrogens are substances that induce fever when introduced into the human body, and they are often associated with the presence of bacterial endotoxins. Controlling and testing pyrogens play a important role in the pharmaceutical and healthcare industries, as they cause fever and other immune responses in patients. In 2023, the pyrogen testing market demonstrated a varied distribution among different test types. The Limulus Amebocyte Lysate (LAL) test emerged as the most dominant method, accounting for 54.13% of the market share. This test, known for its high sensitivity and specificity in detecting endotoxins, is widely preferred in the industry. The Rabbit Pyrogen Test followed with a substantial share of 18.85%. Despite its traditional use and effectiveness, this method is less favored as compared to LAL due to factors such as the ethical considerations and variability in results. In addition, the others segment held a significant 27.02% of the market.
Pyrogen Testing Market Segmentation
The pyrogen testing industry is segmented on the basis of product & service, test type, end user, and region. By product & service, the market is classified into reagents & kits, instruments, and services. By test type, the market is divided into LAL tests, rabbit pyrogen test, and others. By end user, it is segregated into pharmaceutical & biotechnology companies, medical device companies, and others. Region-wise, the market is analyzed across North America, Europe, Asia-Pacific, and LAMEA.
Regional/Country Market Outlook
North America dominated the pyrogen testing market share in 2023 owing to substantial R&D activities, strong presence of major key players, well-established pharmaceutical industry, technological advancements, and stringent regulations and standards imposed by agencies such as the U.S. Food and Drug Administration and U.S. Pharmacopeia. According to pyrogen testing market opportunity analysis in the Asia-Pacific region, rapid market expansion is anticipated due to improving healthcare infrastructure, growing pharmaceutical and biopharmaceutical industry, and rise in drug manufacturing. Surge in drug manufacturing activities and contract manufacturing organizations (CMOs) in Asia-Pacific increases the demand for pyrogen testing to meet both local and international regulatory requirements.
- According to a 2024 article by Invest India, Government of India, India is among the Top 12 destinations for biotechnology globally and 3rd largest destination for biotechnology in Asia-Pacific. India’s BioEconomy has crossed an estimated $130 Bn in 2024 and has witnessed increase in valuation in the past eleven years.
- According to a 2023 fact sheet by Invest India, the hospital sector in India is expected to grow at CAGR of 18.24% from 2021 to 2027.
- According to a 2023 article by Government of UK, businesses in the UK life sciences industry generated $120.85 billion in turnover in 2021/22
How Are Consumer Preferences Shaping Market Trends?
Consumer preferences in the pyrogen testing market demand for higher safety and quality assurance. Patients, healthcare providers, and regulatory bodies prefer manufacturing processes that are adhered to strict regulations. This increases the demand for testing solutions that deliver accurate, reproducible results. Pyrogen testing products that provide reliable endotoxin detection are preferred in biologics, vaccines, and sterile injectable drugs where patient risk is highest.
Rise in demand for ethical, animal-free testing methods boost the demand for pyrogen testing. Consumers prefer in vitro alternatives owing to the associated precision. Consumers also value speed and efficiency in testing workflows. Testing solutions that deliver faster turnaround times without compromising quality are preferred, especially in fast-paced manufacturing environments where batch release timelines can significantly impact production schedules. Advanced testing kits and automated platforms that reduce manual errors and increase throughput are thus gaining traction.
Industry Trends
- According to a 2021 report by the National Library of Medicine, U.S. Pharmacopeia (USP), Japanese Pharmacopeia (JP), and European Pharmacopeia (Ph. Eur.) harmonized three types of Limulus Amebocyte Lysate tests (gel-clot, chromogenic, and turbidimetric techniques) for the purpose of evaluating endotoxin contamination in parenteral drugs, medical devices, and raw materials.
- According to a 2021 report by the U.S. National Science Foundation, an estimated 70 million endotoxin tests are performed each year in the U.S. alone.
- According to a 2021 article by the National Library of Medicine, amebocyte lysate test is the most sensitive and reliable method applied for in vitro detection of bacterial endotoxins.
- According to a 2023 article by National Library of Medicine, the standard pyrogenicity assessing methods required by the European Pharmacopoeia are the rabbit pyrogen test (RPT), the bacterial endotoxin (BE) test (the LAL test), the recombinant factor C test, and the monocyte activation test (MAT).
Who Are the Primary End Users Driving Market Adoption?
The major end users in the pyrogen testing market include pharmaceutical and biotechnology companies. These firms produce a wide range of injectable drugs, vaccines, biologics, and biosimilars, which must be tested for pyrogen contamination before regulatory approval and market distribution. Pyrogen testing plays an important role in quality control activities throughout the drug development and manufacturing process.
Medical device manufacturers also use pyrogen testing. Many medical devices may come into contact with blood, which mandates pyrogen testing to ensure products are free from contaminants. Further, contract research organizations and third-party laboratories also use pyrogen testing. Many companies outsource their pyrogen testing to contract research organizations which helps them save cost, gain expertise, and access advanced equipment without large capital investments.
What Role Do Digital Technologies Play in Improving Products and Services?
Digital technologies play an important role in modernizing the pyrogen testing market. These technologies help improve laboratory efficiency, enhance traceability, and ensure regulatory compliance. They allow laboratories to manage sample data, automate reporting, and maintain accurate records.
Digital platforms also enhance quality assurance processes. Automated documentation ensures that each step of the testing process is recorded accurately. This strengthens product reliability and reduces the risk of compliance issues. Cloud-based systems further help improve collaboration between research, manufacturing, and quality teams.
How Is Artificial Intelligence Transforming This Market?
Artificial intelligence plays an important role in pyrogen testing. AI-powered systems can analyze this data quickly, identify patterns, and detect abnormal results. This reduces human error and ensures more reliable test outcomes. It also supports predictive quality control. Further, AI also helps manage sample tracking, schedule tests, and ensure compliance with regulatory standards. These intelligent systems reduce administrative burden and help laboratories maintain consistent documentation, which is critical for audits and regulatory inspections.
Competitive Landscape
The major players operating in the pyrogen testing market include Merck KGaA, Lonza Group, Thermo Fisher Scientific Inc., FUJIFILM Holdings Corporation, GenScript, SEIKAGAKU CORPORATION, Charles River Laboratories, STERIS, WuXi AppTec, and Eurofins Scientific. Other players in the pyrogen testing market are Fuzhou Xinbei, and Wako Chemicals
What are the Recent Key Strategies and Developments
- In October 2023, Lonza Group launched two new rapid monocyte activation test (MAT) systems, the PyroCell MAT Rapid System and PyroCell MAT Human Serum (HS) Rapid System, to streamline and simplify rabbit-free pyrogen testing.
- In December 2022, Charles River Laboratories International, Inc. launched the Endosafe Nexus 200, expanding its robust endotoxin testing portfolio.
- In December 2021, FUJIFILM Wako Pure Chemical Corporation launched the recombinant protein reagent PYROSTAR Neo for detection of bacterial endotoxin.
What are the Key Benefits for Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the pyrogen testing market analysis from 2025 to 2033 to identify the prevailing pyrogen testing market opportunities.
- The pyrogen testing market size research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the pyrogen testing market segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global pyrogen testing market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global pyrogen testing market trends, key players, market segments, application areas, and market growth strategies.
Pyrogen Testing Market Report Highlights
| Aspects | Details |
| Market Size By 2033 | USD 3.6 Billion |
| Growth Rate | CAGR of 8.3% |
| Forecast period | 2024 - 2033 |
| Report Pages | 280 |
| By Product And Service |
|
| By Test Type |
|
| By End User |
|
| By Region |
|
| Key Market Players | Wuxi AppTec, Merck KGaA, Thermo Fisher Scientific Inc., Steris Corporation, Genscript Technology Corporation, Eurofins Scientific SE, SEIKAGAKU CORPORATION, Lonza Group AG, Charles River Laboratories, FUJIFILM Corporation |
The global pyrogen testing market size was valued at $1.6 billion in 2023
The market value of pyrogen testing market is projected to reach $3.6 billion by 2033
The forecast period for pyrogen testing Market is 2024-2033.
The base year is 2023 in pyrogen testing market
Major key players that operate in the pyrogen testing Market are Merck KGaA, Lonza Group, Thermo Fisher Scientific Inc., and FUJIFILM Holdings Corporation.
Loading Table Of Content...
Loading Research Methodology...



