Thrombectomy Devices Market Statistics - 2030
The global thrombectomy devices market size was valued at $1.3 billion in 2020, and is projected to reach $2.6 billion by 2030, growing at a CAGR of 7.4% from 2021 to 2030. Thrombectomy device is an intracoronary catheter with central aspiration lumen through which the thrombus is extracted. It is used to treat blood clots, which are present in capillaries, veins, and arteries. Surgical thrombectomy is a procedure used to remove blood clot from an artery or vein, which obstructs blood flow. Thrombectomy devices are designed to minimize damage caused to blood vessels during surgical procedure and reduce blood loss. These devices are used for treatment of acute myocardial infraction, peripheral arterial diseases, deep vein thrombosis, pulmonary embolism, and neurovascular thrombosis conditions.
Key factors that drive thrombectomy devices market growth include increase in prevalence of acute myocardial infarction (AMI), peripheral artery disease (PAD), deep vein thrombosis (DVT), and pulmonary embolism (PE). In addition, increase in sedentary lifestyle, surge in unhealthy living conditions & habits of young generation, and rise in geriatric population drive the growth of the thrombectomy devices market size. Increase in incidence of cardiovascular diseases, growth in demand for minimally invasive procedures, surge in initiative by government and private organization for better healthcare infrastructure, and rise in competition among key players led to technological advancements and innovations in thrombectomy devices industry, driving the growth of the thrombectomy devices market share. Meanwhile, increase in number of approvals for new products propel the growth of the market. For instance, in January 2021, the U.S. Food and Drug Administration (FDA), announced clearance for AlphaVac mechanical thrombectomy system, which is an off-circuit, multi-purpose mechanical aspiration thrombectomy device. It is used for non-surgical removal of thrombi or emboli from vasculature.
Moreover, in December 2021, Terumo Aortic, a medical device company, announced launch of aortic balloon device. This device assists physicians in expansion of aorta while using TREO and RELAY stent grafts in endovascular aortic repair. Opportunities in thrombectomy device market are due to introduction of lasers, UV radiation, and 3D imaging generation in thrombectomy devices. Laser facilitated thrombectomy can be another therapeutic option for removal of clots from arteries of brain and heart. In addition, 3D imaging along with thrombectomy is popular in current healthcare industry . Furthermore, combination of decompressive hemicraniectomy and endovascular thrombectomy treatment in patients appeared to be safe and provide good clinical outcome. This factors drive the growth of the thrombectomy device market.
However, unavailability of trained professionals, lack of awareness about thrombectomy devices, and complicated use of thrombectomy devices are expected to restrict the market growth during the thrombectomy devices market forecast period.
Thrombectomy Device Market Segmentation
The thrombectomy devices market is segmented into technology, type, utility, application, and region. According to technology, it is categorized into mechanical thrombectomy devices, aspiration thrombectomy devices, and rheolytic thrombectomy devices. Depending on type, it is divided into automated devices and manual devices. As per the utility, it is bifurcated into reusable devices and disposable devices. By application, it is categorized into peripheral diseases, coronary diseases, and neural diseases.
Region wise, market is analyzed across North America (the U.S., Canada, and Mexico), Europe (Germany, France, the UK, Italy, and rest of Europe), Asia-Pacific (Japan, China, India, Australia, and rest of Asia-Pacific), and LAMEA (Brazil, Saudi Arabia, South Africa, and rest of LAMEA).
Segment review
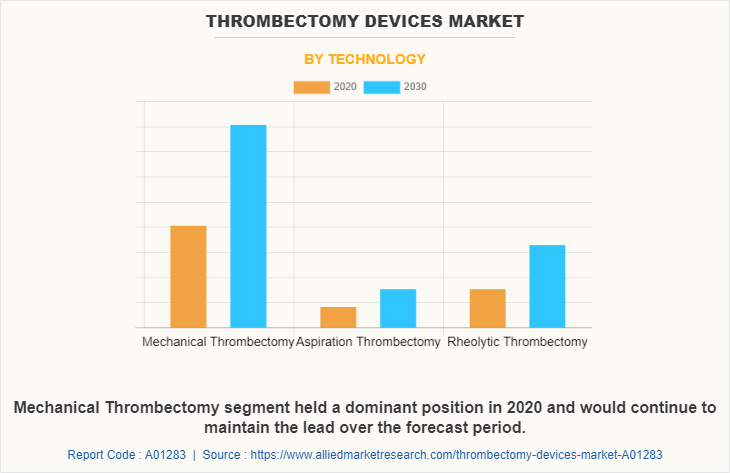
Depending on technology, the mechanical thrombectomy device segment dominated the market in 2020, and this trend is expected to continue during the forecast period, owing to development of new innovative mechanical thrombectomy devices and increase in number of key players for manufacturing of thrombectomy devices.
However, the rheolytic segment is expected to witness considerable growth during the forecast period, owing to launch of novel and more advanced products by key players and advancement in technology in medical device sector.
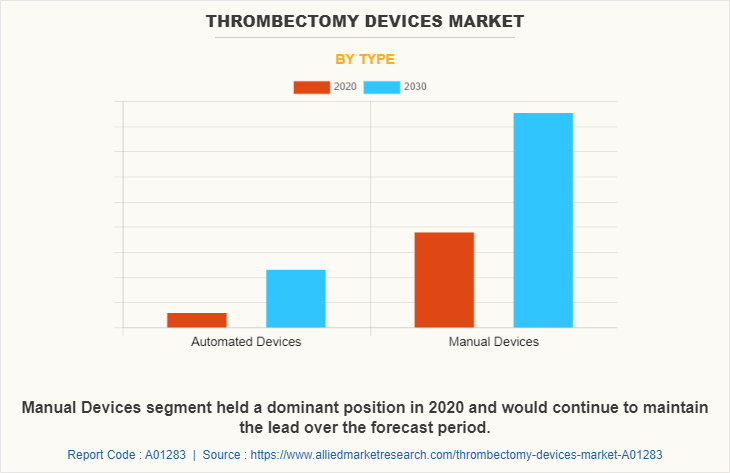
By type, manual thrombectomy device segment dominated the market in 2020, and this trend is expected to continue during the forecast period, owing to increase in R&D activities in healthcare sector and development of advanced thrombectomy device. However, the automated device segment is expected to witness considerable growth during the forecast period, due to increase in its demand.Automated device saves labor and provides precise & accurate results with improved quality. Automated thrombectomy devices use various control systems, which work automatically in removal of clot from blood vessels. These devices include thrombectomy devices, which work sequentially and are controlled by computers.
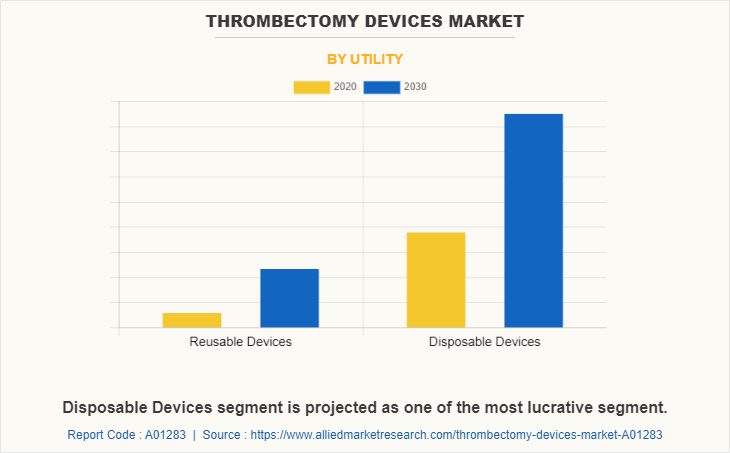
As per utility, disposable thrombectomy device segment dominated the market in 2020, and this trend is expected to continue during the forecast period, owing to increase in use of disposable thrombectomy devices, which limits the infection. However, reusable thrombectomy device segment is expected to witness considerable growth during the forecast period, due to increase in launch of novel product and rise in number of key players for development of reusable thrombectomy devices.
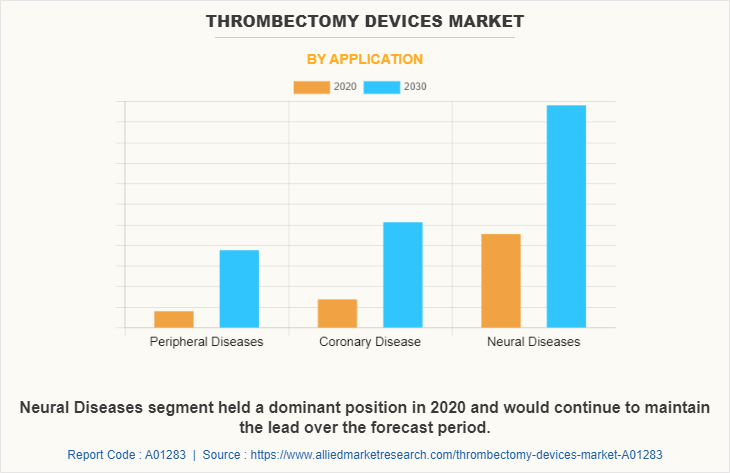
According to application, neural disease segment dominated the market in 2020 and this trend is expected to continue during the forecast period, owing to rise in prevalence of neurological disorder and implementation of government policies to spread awareness about the neural disease among population.
However, coronary disease segment is expected to witness considerable growth during the forecast period, due to increase in prevalence of coronary heart disease, rise in geriatric population, and advancement in R&D activities for the treatment of coronary artery disease.
Region-wise, North America dominated the thrombectomy device market in 2020 and is expected to be dominant during the forecast period, owing to surge in advanced health care facility, rise in prevalence of neurological disorder, increase in number of approval for thrombectomy devices, growth in number of surgical procedure, and rise in presence of key players. In addition, the U.S. is highly affected by cardiovascular diseases and acute ischemic stroke. However, Asia-Pacific is expected to register highest CAGR of 8.8% from 2021 to 2030, owing to large patient population, increase in government insurance coverage in developing countries, rise in approval of new innovative embolization, and mechanical thrombectomy products.
The key players operating in the global thrombectomy device market include, AngioDynamics, Inc., Johnson & Johnson, Inc., Penumbra Inc., Terumo Corporation, Abbott, Boston Scientific Corporation, Medtronic plc., Stryker Corporation, and Teleflex Incorporated.
Key Benefits For Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the thrombectomy devices market analysis from 2020 to 2030 to identify the prevailing thrombectomy devices market opportunity.
- The market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the thrombectomy devices market segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global thrombectomy devices market trends, key players, market segments, application areas, and market growth strategies.
Thrombectomy Devices Market Report Highlights
| Aspects | Details |
| By Technology |
|
| By Type |
|
| By Utility |
|
| By Application |
|
| By Region |
|
| Key Market Players | Penumbra Inc., Terumo Corporation, Teleflex Incorporated, Medtronic plc, Abbott Laboratories, AngioDynamics, Inc, Boston Scientific Group, Johnson and Johnson, Stryker Corporation, Koninklijke Philips N.V. |
Analyst Review
In accordance with the various opinions of top-level CXOs in the thrombectomy devices industry, utilization of thrombectomy devices is expected to witness a significant rise with increase in treating neurovascular, peripheral, and coronary disorders. The market for thrombectomy devices piqued the interest of healthcare, due to the fact that it is used to treat clots efficiently. Devices, including mechanical, rheolytic, and aspiration thrombectomy products are used to treat peripheral, coronary, and neural clot related diseases. Remarkable increase in usage of thrombectomy devices products for the management of chronic disease patients propels the growth of the market. In addition, increase in incidence of acute myocardial infarction (AMI), peripheral artery disease (PAD), deep vein thrombosis (DVT), and pulmonary embolism (PE), due to sedentary lifestyle, unhealthy living conditions, drive the thrombectomy device market growth. North America and Europe are expected to offer high growth opportunities to the key players.
According to perspectives of the CXOs of leading companies in the market rise in geriatric population, increase in demand for minimally-invasive procedures, technological advancements in medical device sector, and surge in innovations by key players drive the growth of the market. However, unavailability of trained professionals, lack of awareness about thrombectomy devices, and complicated use of thrombectomy devices restrain the market growth. presently, mechanical thrombectomy devices is the largest segment of the thrombectomy devices market. According to the CXOs, increase in incidence of cardiovascular diseases and need for better healthcare infrastructure fuel the growth of the market. As per CXOs, North America is expected to dominate the thrombectomy devices market during the forecast period.
The total market value of thrombectomy devices market is $1,269 million in 2020.
The forecast period in the report is from 2021 to 2030
The market value of thrombectomy devices Market in 2021 was $1,351.5 million
The base year for the report is 2020.
Yes, thrombectomy devices companies are profiled in the report
The top companies that hold the market share in thrombectomy devices market are AngioDynamics, Inc., Johnson & Johnson, Inc., Penumbra Inc., Terumo Corporation, Abbott, Boston Scientific Corporation, Medtronic plc., Stryker Corporation, and Teleflex Incorporated
The key trends in the thrombectomy devices market are increase in prevalence of acute myocardial infarction (AMI), increase in sedentary lifestyle, surge in unhealthy living conditions & habits of young generation, and rise in geriatric population drive the growth of the market
Loading Table Of Content...



