Vaccine Adjuvants Market Research, 2035
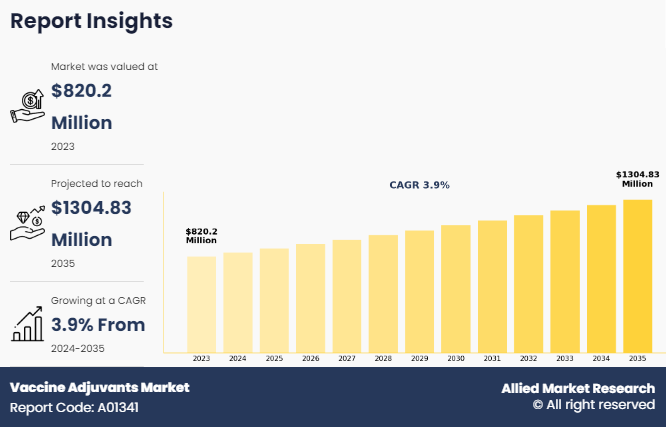
The global vaccine adjuvants market was valued at $820.2 million in 2023, and is projected to reach $1,304.83 million by 2035, growing at a CAGR of 3.9% from 2024 to 2035. The vaccine adjuvants market growth is driven by increase in demand for effective vaccines to combat infectious diseases and rise in awareness about immunization programs globally. For instance, according to the Centers for Disease Control and Prevention (CDC), public health laboratories tested 129,638 specimens and identified 39,885 as influenza positive during 2023–2024 in the U.S. Of these, 30,689 (76.9%) were influenza A, and 9,196 (23.1%) were influenza. This highlights the ongoing need for effective vaccines and adjuvants to address the significant prevalence of influenza and other infectious diseases.
Vaccine adjuvants are substances added to vaccines to enhance the body's immune response to the antigen, helping to produce a stronger and longer-lasting immunity. They work by stimulating the immune system to recognize and respond more effectively to the vaccine's active ingredients. Adjuvants play a crucial role in reducing the amount of antigen required per dose, improving vaccine efficacy, and sometimes enabling protection against multiple strains of a pathogen. Commonly used adjuvants include aluminum salts and formulations such as oil-in-water emulsions, which are designed to meet the needs of modern vaccines targeting a variety of infectious and non-infectious diseases.
Key Takeaways
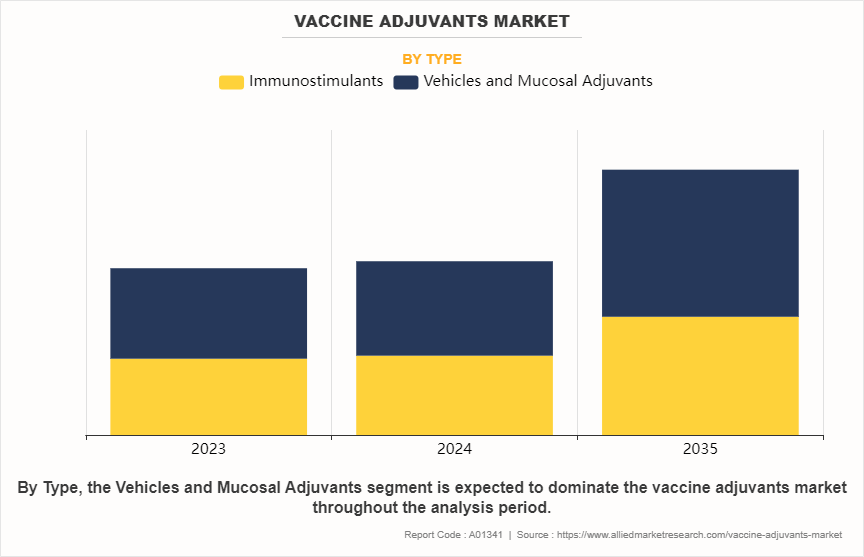
- By type, the vehicles and mucosal adjuvants segment dominated the global vaccine adjuvants market size in 2023 and is expected to register the highest CAGR during the forecast period.
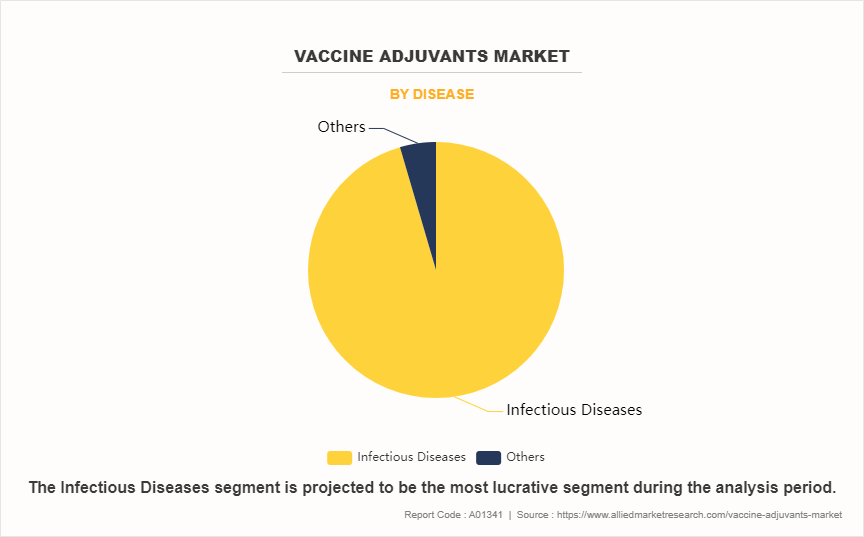
- By disease, the infectious diseases segment dominated the global market in terms of revenue in 2023 and is expected to register the highest CAGR during the forecast period.
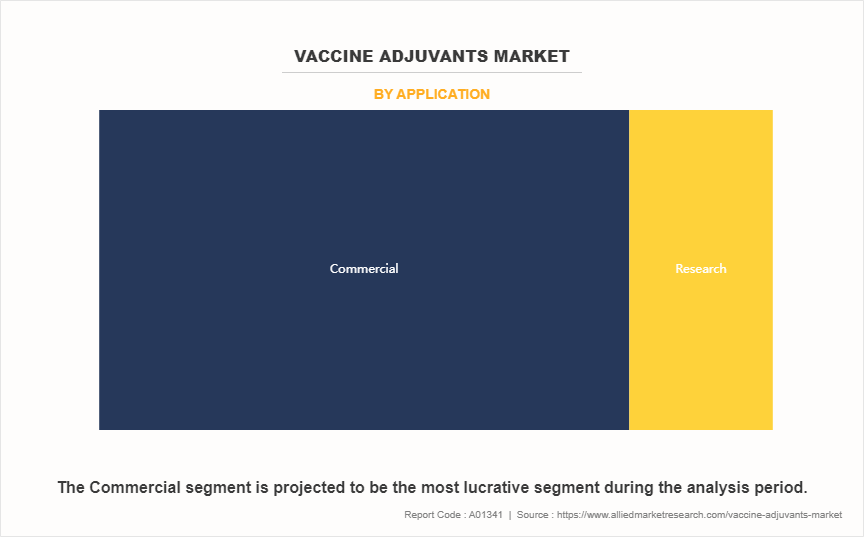
- By application, the commercial segment dominated the global market in terms of revenue in 2023 and is also expected to register the highest CAGR during the forecast period.
- By region, North America dominated the vaccine adjuvants market share in terms of revenue in 2023. However, Asia-Pacific is expected to register the highest CAGR during the forecast period.
Market Dynamics
The vaccine adjuvants market growth is driven by increase in prevalence of infectious diseases and rise in awareness about immunization programs globally. Vaccine adjuvants are crucial components of vaccines that enhance the immune response, making them indispensable in developing more effective immunization strategies. Their ability to boost immune reactions has led to their adoption in vaccines targeting various infectious diseases, cancers, and even allergies.
One of the primary drivers of this vaccine adjuvants market forecast is rise in prevalence of infectious diseases globally. In addition, rise in advancements in adjuvant formulations has played a key role in the market growth. New-generation adjuvants, such as MF59 (an oil-in-water emulsion) and AS04 (a combination of aluminum hydroxide and a TLR4 agonist), have demonstrated improved safety profiles and efficacy in clinical trials. These advancements expanded the scope of adjuvants, enabling their use in vaccines for diseases such as COVID-19, malaria, and human papillomavirus (HPV).
Furthermore, government initiatives and funding for vaccine research and development significantly supporte the growth of the vaccine adjuvants market. For instance, organizations such as Gavi, the Vaccine Alliance, National Institute of Health (NIH), and the World Health Organization (WHO) actively promote vaccination programs globally, boosting the demand for advanced vaccines with adjuvants.
However, high investment and R&D costs associated with vaccine adjuvants, and regulatory & safety concerns are expected to pose hindrances to the widespread adoption of vaccine adjuvants, thereby restraining the market growth. In addition, although most adjuvants are safe and well-tolerated, some can cause side effects such as local inflammation or allergic reactions. These safety concerns can lead to hesitancy among healthcare providers and the public, potentially impacting the adoption of vaccines containing adjuvants and restricts the vaccine adjuvants market opportunity.
On the other hand, the vaccine adjuvants market expanded significantly in emerging markets owing to the development of healthcare infrastructure and increased R&D activities. Rapid urbanization and economic growth in countries such as China and India have created substantial opportunities for vaccine adjuvant manufacturers to cater to the needs of these growing markets. Government and private sector funding for vaccine research and development also play a crucial role in expanding opportunities in the vaccine adjuvants market. Government initiatives and partnerships with pharmaceutical companies foster innovation and enable the creation of vaccines for underserved populations and diseases with unmet needs.
Segments Overview
The vaccine adjuvants market is segmented into type, disease, application, and region. By type, it is classified into immunostimulants and vehicles and mucosal adjuvants. On the basis of disease, the market is categorized into infectious diseases and others. On the basis of application, the market is bifurcated into research and commercial. Region-wise, the market is analyzed across North America, Europe, Asia-Pacific, Latin America, and Middle East and Africa.
By Type
The vehicles and mucosal adjuvants segment dominated the vaccine adjuvants market size in 2023 and is anticipated to register the fastest CAGR growth during the forecast period. This is attributed to their ability to enhance antigen stability, improve immune response, and facilitate targeted delivery. In addition, rise in demand for advanced vaccine formulations and growing prevalence of infectious diseases are driving the adoption of vehicles and mucosal adjuvants in vaccine development.
By Disease
The infectious disease segment dominated the market share in 2023 and is anticipated to register the fastest CAGR growth during the forecast period. This is attributed to increase in prevalence of infectious diseases globally, growing vaccination programs, and rise in focus on preventive healthcare. In addition, advancements in vaccine technology and development of novel vaccines targeting a broader range of pathogens further support the growth of this segment.
By Application
The commercial segment held the largest vaccine adjuvants market share in 2023 and is anticipated to register the fastest CAGR growth during the forecast period. This is attributed to increase in adoption of adjuvants in large-scale vaccine production, driven by rise in demand for vaccines against infectious diseases and chronic conditions. Moreover, advancements in manufacturing technologies and strategic collaborations between pharmaceutical companies and research organizations are further fueling the expansion of this segment.
By Region
The vaccine adjuvants industry is analyzed across North America, Europe, Asia-Pacific, Latin America, and Middle East Africa. North America dominated the market share in 2023. This dominance is attributed to the presence of leading vaccine manufacturers, advanced healthcare infrastructure, and significant investment in research and development activities. In addition, strong government support for immunization programs, rise in prevalence of infectious and chronic diseases, and increase in public awareness regarding vaccination contribute to the market growth in the region.
However, the Asia-Pacific region is anticipated to register the highest CAGR during the forecast period. This is attributed to the growing population, increase in prevalence of infectious diseases, and rise in healthcare expenditure in emerging economies such as China and India. In addition, government initiatives to improve immunization coverage, expanding pharmaceutical manufacturing capabilities, and increasing investments in vaccine research and development are driving the market growth in the region.

Competitive Analysis
Key players such as GlaxoSmithKline plc., and Croda International Plc. have adopted agreement, partnership, and product approval as key developmental strategies to improve the product portfolio of the vaccine adjuvants market. For instance, in March 2024, Croda International Plc (Croda) and The Access to Advanced Health Institute (AAHI) signed partnership agreement around innovation and development of adjuvant formulations, under which Croda and AAHI will leverage their combined expertise to make novel vaccine adjuvant formulations globally accessible, thereby advancing next generation, robust and durable vaccines.
Recent Development in the Vaccine Adjuvants Industry
- In August 2024, GSK plc announced that the European Commission authorized Arexvy (respiratory syncytial virus vaccine, recombinant adjuvanted) for active immunization for the prevention of lower respiratory tract disease (LRTD) caused by respiratory syncytial virus (RSV) in adults aged 50-59 who are at increased risk.
- In October 2024, GSK plc announced new data from the AReSVi-006 phase III trial evaluating the efficacy of a single dose of Arexvy (respiratory syncytial virus vaccine, recombinant adjuvanted) against lower respiratory tract disease (LRTD) caused by respiratory syncytial virus (RSV) in adults aged 60 years and older, including those at increased risk over three full RSV seasons (NCT04886596).
- In March 2023, Aurorium announced the acquisition of CENTAURI Technologies. The addition of CENTAURI’s complementary specialty material product portfolio and advanced manufacturing capabilities expands Aurorium’s offering and extends its reach in high-growth end markets.
- In February 2021, Dynavax Technologies Corporation announced that the European Commission has granted Marketing Authorization for HEPLISAV B (Hepatitis B Vaccine (Recombinant), Adjuvanted) for the active immunization against hepatitis B virus infection (HBV) caused by all known subtypes of hepatitis B virus in adults 18 years of age and older.
Key Benefits For Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the vaccine adjuvants market analysis from 2023 to 2035 to identify the prevailing vaccine adjuvants market opportunities.
- The market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the vaccine adjuvants market segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global vaccine adjuvants market trends, key players, market segments, application areas, and market growth strategies.
Vaccine Adjuvants Market Report Highlights
| Aspects | Details |
| Market Size By 2035 | USD 1.3 billion |
| Growth Rate | CAGR of 3.9% |
| Forecast period | 2023 - 2035 |
| Report Pages | 339 |
| By Type |
|
| By Disease |
|
| By Application |
|
| By Region |
|
| Key Market Players | Adjuvatis, Croda International Plc, InvivoGen, Aurorium, Dynavax Technologies Corporation, GlaxoSmithKline plc., Novovax Inc., Agenus Inc., Associated British Foods, Air Liquide Group |
Analyst Review
This section provides various opinions of top-level CXOs in the global vaccine adjuvants market. According to the insights of CXOs, the global vaccine adjuvants market is expected to exhibit high growth potential attributable to increase in demand for more effective vaccines to combat infectious diseases. Advancements in vaccine technology,including the development of next-generation adjuvants, are further driving market expansion. Further, rise in investments in research and development and growing public health initiatives worldwide contribute to the market growth. The focus on improving vaccine efficacy and safety also enhances the market growth.
CXOs further added that the growing prevalence of infectious diseases, coupled with the need for enhanced immune responses, is a key driver for the adoption of advanced vaccine adjuvants. They emphasized that public-private partnerships, increasing healthcare investments, and the expansion of vaccination programs in emerging markets further fuel demand for vaccine adjuvants. In addition, innovations in adjuvant formulations, such as mucosal and nanoparticle-based technologies, present significant opportunities for the market growth.
Furthermore, North America dominated the market share, in terms of revenue in 2023, owing to surge in cases of infectious diseases, cancer, high healthcare expenditure, presence of major biopharmaceutical companies, and a strong focus on vaccine innovation, along with favorable government policies supporting vaccination initiatives. However, Asia-Pacific is anticipated to witness notable growth owing to a large and diverse target population, rise in healthcare expenditures, and increase in government initiatives to enhance immunization programs. The growing prevalence of infectious diseases, coupled with advancements in biotechnology and vaccine manufacturing capabilities, is driving demand for more effective adjuvants. In addition, the region's expanding pharmaceutical sector and rise in awareness of preventive healthcare, present significant opportunities for the vaccine adjuvants market growth.
The total market value of vaccine adjuvants market was $820.2 million in 2023.
The forecast period for vaccine adjuvants market is 2024 to 2035.
The market value of vaccine adjuvants market is projected to reach $1,304.83 million by 2035.
The base year is 2023 in vaccine adjuvants market .
Top companies such as GlaxoSmithKline plc., and Croda International Plc. held a high market position in 2023.
The growth of the vaccine adjuvants market is driven by the increase in demand for enhanced vaccine efficacy, particularly for emerging infectious diseases. Additionally, rising government investments and funding for vaccine research and development are supporting market expansion.
The vehicles and mucosal adjuvants segment holds the largest market share in 2023. This is attributed to their ability to enhance the stability and delivery of active vaccine ingredients. These adjuvants are widely used for their cost-effectiveness and compatibility with various vaccine formulations. Their proven track record in improving immune responses drives their continued dominance in the market.
Vaccine adjuvants are substances added to vaccines to enhance the body's immune response to the vaccine's active ingredient, typically an antigen. They help stimulate a stronger, longer-lasting immunity, often requiring a smaller dose of the antigen.
Loading Table Of Content...
Loading Research Methodology...



