Deferiprone Market Summary
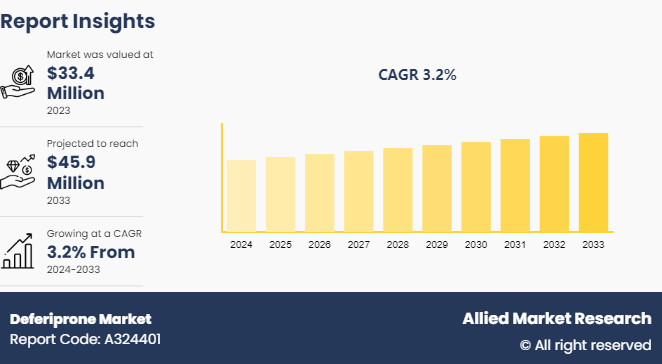
The global deferiprone market was valued at $33.4 million in 2023, and is projected to reach $45.9 million by 2033, growing at a CAGR of 3.2% from 2024 to 2033. Rise in prevalence of iron overload disorders, surge in awareness about rare hematological diseases, and increase in adoption of oral chelation therapies are the major factors which drive the deferiprone market growth.
Market Introduction and Definition
The Deferiprone market is the comprehensive ecosystem of pharmaceutical products, research activities, and distribution networks focused on the development, production, and commercialization of deferiprone-based therapies. Deferiprone is an oral iron chelator primarily used for the treatment of transfusional iron overload in patients with conditions such as thalassemia syndromes and other chronic anemias requiring regular blood transfusions. The market encompasses branded and generic formulations, hospital and retail pharmacy distribution channels, and ongoing clinical research aimed at expanding therapeutic indications.
The deferiprone market plays a significant role in managing iron overload, a serious complication that can lead to organ damage affecting the heart, liver, and endocrine system. Deferiprone helps reduce morbidity and improve long-term patient outcomes by binding excess iron and promoting its excretion. Growing diagnosis rates of beta-thalassemia and sickle cell disease, particularly in emerging economies, are contributing to increased demand for effective iron chelation therapies.
Technological advancements in drug formulation, improved safety monitoring protocols, and supportive regulatory frameworks for orphan drugs have strengthened market development. In addition, increasing healthcare expenditure, expanding patient access programs, and rising investment in rare disease research further support market expansion. Continuous clinical studies evaluating combination therapies and broader applications are expected to create new growth opportunities for the global deferiprone market.
Key Takeaways
- The deferiprone market size covers 20 countries. The research includes a segment analysis of each country in terms of value ($Million) for the projected period 2024-2033.
- More than 1, 500 product literatures, industry releases, annual reports, and other such documents of major deferiprone industry participants along with authentic industry journals, trade associations' releases, and government websites have been reviewed for generating high-value industry insights.
- The study integrated high-quality data, professional opinions and analysis, and critical independent perspectives. The research approach is intended to provide a balanced view of global markets and to assist stakeholders in making educated decisions in order to achieve their most ambitious growth objectives.
Key Market Dynamics
The deferiprone market is evolving steadily, supported by increasing awareness of iron overload management, growing diagnosis of rare hematological disorders, and advancements in oral chelation therapies.
Drivers
Rising prevalence of transfusion-dependent conditions such as beta-thalassemia, sickle cell disease, and other chronic anemias is a major factor driving the growth of the deferiprone market. Regular blood transfusions in these patients often lead to iron overload, which can cause severe complications including cardiac, hepatic, and endocrine dysfunction. Growing screening programs and improved diagnostic capabilities are enabling early detection and timely treatment, thereby supporting consistent demand for iron chelation therapies.
Increasing preference for oral chelators over parenteral alternatives further accelerates market growth. Deferiprone offers convenient oral administration and effective myocardial iron removal, making it a valuable therapeutic option. Supportive regulatory frameworks for orphan drugs, favorable clinical guidelines, and expanding patient assistance programs also strengthen adoption. Additionally, rising healthcare expenditure in emerging economies and improved access to specialized hematology centers contribute to market expansion.
Opportunities
Ongoing clinical research exploring combination therapies and expanded indications presents significant growth opportunities. Investigations into deferiprone use in conditions such as neurodegenerative disorders and other iron-related pathologies may broaden its therapeutic scope. Advancements in formulation development, including improved safety monitoring strategies and extended-release versions, create further opportunities for manufacturers. Increasing collaborations between pharmaceutical companies and research institutions also support innovation and pipeline development.
Restraints
Strict safety monitoring requirements, including the risk of agranulocytosis and neutropenia, may limit widespread adoption. High treatment costs and limited awareness in low-income regions can restrict patient access. In addition, stringent regulatory approvals and small patient populations associated with rare diseases may pose challenges to sustained market growth during the forecast period.
Market Segmentation
The deferiprone market is segmented into form, application, distribution channel, and region. On the basis of form, the market is categorized into tablets and others. On the basis of application, the market is divided into transfusional iron overload, and NTDT Caused Overload. On the basis of distribution channel, the market is categorized into hospital pharmacies, drug store & retail pharmacies and online providers. Region wise, the market is analyzed across North America, Europe, Asia-Pacific, and LAMEA.
Regional/Country Market Outlook
North America accounted for a major deferiprone market size of the deferiprone market owing to the presence of several major players, high prevalence of thalassemia, sickle cell disease, well-established healthcare infrastructure, and rising awareness and diagnosis.
Asia-Pacificdeferiprone market share is estimated to grow with the highest CAGR during the forecast period, owing to the lucrative opportunities for the deferiprone market key players, owing to a rise in target population, and awareness about iron overload disorders. Another factor driving the growth of the market is the focus of major players on generic drug availability in deferiprone.
Industry Trends
- Ongoing research and development in the field of iron chelation are leading to improved formulations and combination therapies that enhance the efficacy and safety profile of deferiprone.
- Recent regulatory approvals in various regions and the expansion of indications for deferiprone use are contributing to market growth. Regulatory bodies are recognizing the importance of addressing iron overload, leading to broader access to deferiprone.
Key End-User Segments Fueling Market Growth
The deferiprone market is driven by several primary end users. Hospitals represent the largest end-user segment, serving as the main centers for diagnosis and management of transfusion-dependent disorders such as beta-thalassemia and sickle cell disease. Hematology departments within tertiary care hospitals administer comprehensive treatment plans that include iron chelation therapy, routine blood transfusions, and continuous monitoring of iron levels. Physicians and specialists rely on established treatment protocols and safety monitoring systems to optimize deferiprone therapy and improve long-term patient outcomes.
Specialty hematology and oncology clinics also contribute significantly to market demand. These centers focus on chronic anemia management and provide personalized treatment strategies for patients requiring long-term transfusion support. The convenience of oral deferiprone enhances adherence in outpatient settings, making it a preferred option in specialized clinics. Growing establishment of dedicated rare disease centers and improved access to diagnostic testing further strengthen adoption across this segment.
Retail and hospital pharmacies play a vital role in the distribution and accessibility of deferiprone therapies. As an oral medication, deferiprone is commonly dispensed through hospital pharmacies for inpatients and through retail or specialty pharmacies for outpatient use. Expanding pharmaceutical distribution networks and patient assistance programs improve therapy access, particularly in emerging markets where treatment availability has historically been limited.
Academic institutions and research organizations are another important segment supporting market growth. These institutions conduct clinical trials, pharmacovigilance studies, and research on expanded indications for deferiprone, including combination regimens and potential applications in other iron-related disorders. Government agencies and non-profit organizations focused on rare diseases also collaborate with pharmaceutical companies to enhance awareness and screening initiatives.
In addition, patient advocacy groups and specialized transfusion centers contribute indirectly by promoting early diagnosis, treatment adherence, and comprehensive disease management, thereby supporting sustained demand within the global deferiprone market.
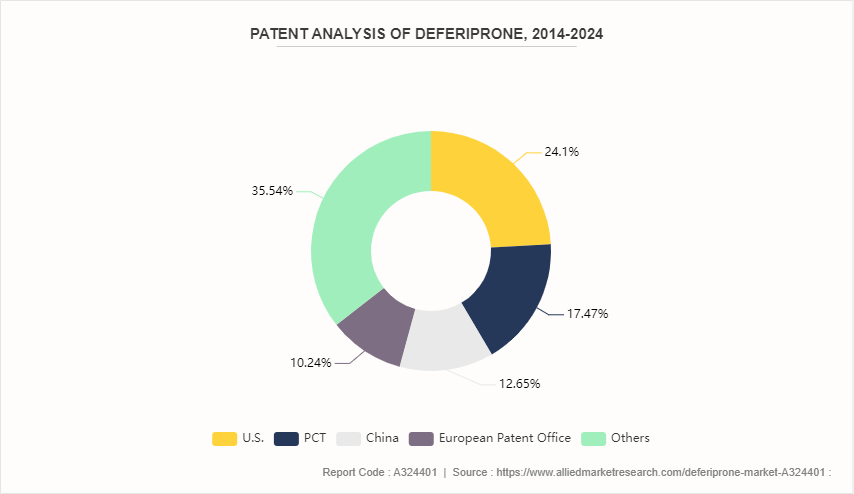
Patent Analysis of Deferiprone, 2014-2024
Although the growth of the deferiprone industry has been more rapid in recent years, the industry can draw on the patents filed by various companies. For instance, patents filed by the U.S. dominated the same with 24.1% share, China holds a share of about 12.7% and PCT holds a share of about 17.5%.
Impact of Evolving Consumer Preferences on Deferiprone Market Trends
Consumer preferences are playing a pivotal role in transforming trends within the deferiprone market. One of the most significant shifts is the growing preference for oral iron chelation therapies over parenteral alternatives. Patients with transfusion-dependent disorders increasingly seek convenient treatment options that fit into daily routines without frequent hospital visits. As a result, orally administered deferiprone is gaining wider acceptance due to ease of use, improved mobility, and better quality-of-life outcomes compared to infusion-based therapies.
Consumers are also prioritizing safety, tolerability, and personalized treatment approaches. There is rising awareness regarding potential adverse effects such as neutropenia and agranulocytosis, prompting patients and caregivers to favor therapies supported by structured monitoring programs and clear clinical guidelines. Healthcare providers are responding by implementing regular blood count monitoring systems and patient education initiatives, strengthening confidence in deferiprone therapy. Transparent communication regarding risk management further influences treatment decisions and brand preference.
Another key trend is the increasing demand for long-term disease management solutions. Patients and families affected by rare hematological disorders are seeking comprehensive care strategies that include access to affordable medications, counseling, and adherence support programs. This drives pharmaceutical companies to expand patient assistance initiatives and improve distribution networks, particularly in emerging economies.
Digital integration is further shaping market dynamics. Patients value teleconsultations, electronic health records, and digital reminder systems that enhance medication adherence and follow-up monitoring. Online patient communities and advocacy platforms also contribute to greater awareness about iron overload management and available treatment options. Additionally, growing sensitivity to drug affordability, insurance coverage, and reimbursement policies influences therapy adoption and provider selection. Collectively, these evolving preferences continue to redefine access strategies, safety frameworks, and innovation pathways across the global deferiprone market.
Competitive Landscape
The major players operating in the deferiprone market include Apotex Inc, Cipla Limited, VHB Life Sciences Limited, Taro Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., Novartis International AG, Chiesi Farmaceutici S.p.A, Zydus Cadila, Cipla Inc., and Pfizer.
Leading Companies and Strategic Initiatives Shaping the Deferiprone Market
Key players operating in the deferiprone market include Apotex Inc., Cipla Limited, VHB Life Sciences Limited, Taro Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., Novartis International AG, Chiesi Farmaceutici S.p.A, Zydus Cadila, Cipla Inc., and Pfizer. These companies maintain strong positions through established hematology portfolios, global distribution capabilities, and sustained investments in rare disease therapeutics.
A central strategy among leading firms is expansion within the orphan drug and specialty pharmaceutical segments. Companies such as Novartis International AG and Chiesi Farmaceutici S.p.A emphasize research-driven development, regulatory approvals, and lifecycle management strategies to strengthen their presence in iron chelation therapy. Meanwhile, Apotex Inc., Cipla Limited, and Sun Pharmaceutical Industries Ltd. focus on cost-effective generic formulations to improve accessibility in emerging and price-sensitive markets.
Strategic partnerships, licensing agreements, and regional expansions are widely adopted to enhance market penetration. Pfizer and Taro Pharmaceutical Industries Ltd. leverage strong international supply chains and compliance frameworks to support global distribution. Zydus Cadila and VHB Life Sciences Limited prioritize expanding manufacturing capacities and strengthening domestic and export networks. Collectively, these strategic initiatives—including product differentiation, regulatory alignment, and patient access programs—reinforce competitive positioning and drive sustained growth across the global deferiprone market.
Recent Key Strategies and Developments in Deferiprone Market
- In recent years, Chiesi Farmaceutici S.p.A has strengthened its rare disease portfolio through continued regulatory expansions and lifecycle management strategies for deferiprone formulations. The company has focused on expanding geographic approvals and enhancing pharmacovigilance programs to reinforce patient safety and long-term treatment adherence in iron overload management.
- Apotex Inc. and Sun Pharmaceutical Industries Ltd. have emphasized the expansion of generic deferiprone offerings across emerging markets. These initiatives aim to improve affordability and increase patient access to oral iron chelation therapy, particularly in regions with a high prevalence of transfusion-dependent thalassemia.
- Cipla Limited has focused on strengthening its hematology portfolio by enhancing distribution partnerships and patient assistance programs to support long-term therapy adherence.
- In addition, Novartis International AG continues to invest in rare disease research collaborations and post-marketing surveillance studies, supporting innovation and reinforcing its strategic presence within the global deferiprone market.?
Key Sources Referred
- National Center for Biotechnology Information
- World Health Organization (WHO)
- National Library of Medicine
- The United States Food and Drug Administration
- U.S. Department of Health & Human Services
- Centers for Disease Control and Prevention
Key Benefits For Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the deferiprone market analysis from 2024 to 2033 to identify the prevailing deferiprone market opportunities.
- The deferiprone market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders to make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the deferiprone market segmentation assists to determine the prevailing market opportunities and deferiprone market forecast.
- Major countries in each region are mapped according to their revenue contribution to the global deferiprone market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global deferiprone market trends, key players, market segments, application areas, and market growth strategies.
Deferiprone Market , by Form Report Highlights
| Aspects | Details |
| Market Size By 2033 | USD 45.9 Million |
| Growth Rate | CAGR of 3.2% |
| Forecast period | 2024 - 2033 |
| Report Pages | 280 |
| By Form |
|
| By Application |
|
| By Distribution Channel |
|
| By Region |
|
| Key Market Players | Chiesi Farmaceutici S.p.A, Taro Pharmaceutical Industries Ltd., VHB Life Sciences Limited, Cipla Inc., Pfizer, Zydus Cadila, Apotex Inc, Sun Pharmaceutical Industries Ltd., Cipla Limited, Novartis International AG |
Increase in iron overload disorders is the upcoming trends of Deferiprone Market in the globe.
Transfusional Iron Overload is the leading application of Deferiprone Market.
North America is the largest regional market for Deferiprone Market.
The deferiprone market was valued at $33.37 million in 2023 and is estimated to reach $45.90 million by 2033, exhibiting a CAGR of 3.2% from 2024 to 2033.
The major players operating in the deferiprone market include Apotex Inc, Cipla Limited, VHB Life Sciences Limited, Taro Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., Novartis International AG, Chiesi Farmaceutici S.p.A, Zydus Cadila, Cipla Inc., and Pfizer.
Loading Table Of Content...



