Stroke Management Market Research, 2032
The global stroke management market was valued at $36.1 billion in 2022, and is projected to reach $74 billion by 2032, growing at a CAGR of 7.4% from 2022 to 2032.The stroke management market growth is driven by advancement in stroke management technology, surge in ageing population, and rise in awareness about early disease diagnosis. Technological advancement in stroke diagnostics techniques is a major driver for the growth of Stroke Management Market Size Such advanced techniques allow accurate examination of the brain and facilitate high resolution of images. Thus, the adoption of technologically advanced diagnostic equipment drives the market growth.
Furthermore, increase in the geriatric population who is more susceptible to stroke also drives the market growth. For instance, according to Medical University of South Carolina (MUSC), about 75% of strokes occur in people aged 65 or older. In addition, according to the World Stroke Organization (WSO) 2022, every year more than 62% of strokes occur in people under 70 years of age. As the incidence of stroke is on the rise, there is an increasing demand for stroke diagnostics and therapeutics, which, in turn drives the market growth.

Key Takeaways:
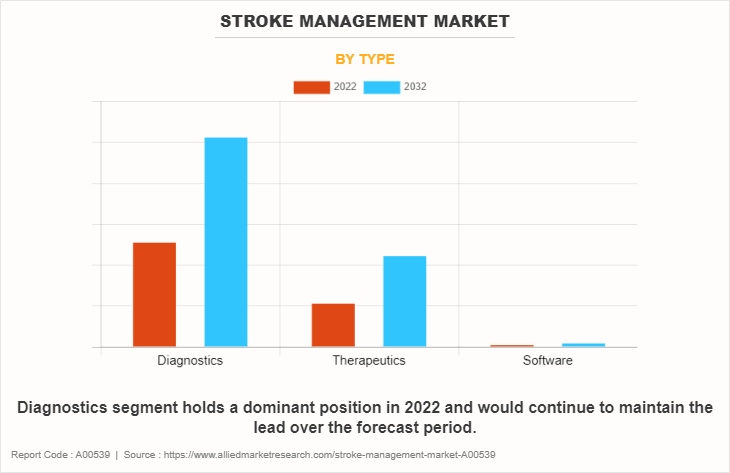
- By type, the diagnostics segment dominated the global market in 2022.
- The software segment is anticipated to be the fastest-growing segment during the forecast period.
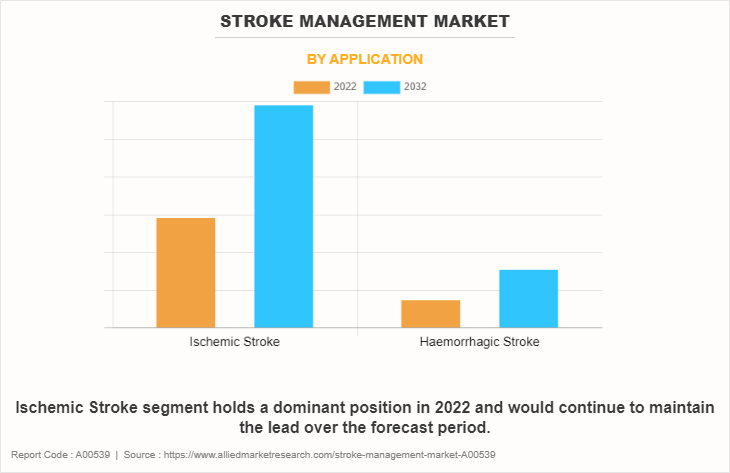
- By application, the ischemic stroke segment dominated the global market in 2022.
- The North America region dominated the market in terms of revenue in 2022.
- Asia-Pacific region is expected to register the highest CAGR during the forecast period.
The global economic recession has a moderate impact on the Stroke Management Industry. The development and accessibility of new diagnostics was impacted by supply chain disruptions, high cost of raw materials, and decrease in consumer spending. However, surge in stroke incidences, increase in patient awareness and development of healthcare in developing countries positively impact the growth of Stroke Management Market Size.
Stroke management describes the healthcare services and treatment given to those who have experienced a stroke. A stroke is a medical condition that occurs when the blood supply to the brain is abruptly cut off, causing brain tissue to be damaged. The management of stroke aims to reduce brain damage, avoid complications, and speed up recovery. Stroke management involves assessment diagnosis, treatment, surgical intervention, and stroke rehabilitation. There are various diagnostic tests such as CT scans, MRI, and blood tests, to determine the type and severity of stroke. Stroke treatment incudes the administration of anticoagulants, tissue plasminogen activators, antihypertensive medications, and antiplatelet drugs. Stroke rehabilitation includes physical therapies to improve quality of life.
Market Dynamics
The growth of stroke management market is driven by increase in incidences of stroke, surge in adoption of telemedicine for stroke management, and rise in government initiatives. The rise in incidence of strokes is a major driving factor in the growth of the stroke management market. Strokes are becoming more common, and this has significant implications for healthcare systems and the demand for effective stroke management. For instance, according to the World Stroke Organization (WSO) 2022, more than 12.2 million new stroke cases are diagnosed every year. As per the same source, 53% of all strokes occur in men. Further, lifestyle-related risk factors also play a substantial role in the increasing incidence of stroke. For instance, sedentary lifestyles, poor dietary choices, and an increase in obesity are contributing to a higher risk of stroke. Thus, increase in incidences of strokes drives the demand for stroke management solutions, which in turn, boosts the market growth.
However, high cost of stroke diagnostics and therapeutics, limited access in low-income countries, and dearth of skilled neurologists restrain the growth of the stroke management market. High costs of stroke diagnosis and treatment create barriers that restrict their adoption in smaller facilities. Thus, limited access to costly stroke management products hampers the market growth.
On the other hand, technological advancement in stroke management and increase in new product approvals are expected to create lucrative opportunities for the market growth during the forecast period. For instance, in July 2022, Siemens Healthineers, a medical technology company has received the Food and Drug Administration (FDA) clearance of the ARTIS icono ceiling, a ceiling-mounted angiography system. This system is designed for a wide range of routine and advanced procedures in interventional radiology and cardiology. In addition, in June 2021, Viz.ai, a healthcare company, received a CE mark for its AI powered stroke care software from European Health, Safety and Environmental Protection Standards. Thus, development of technologically advanced software and diagnostic systems are expected to create lucrative opportunities for the growth of the Stroke Management Industryduring the forecast period.
Segmental Overview
The stroke management market is segmented on the basis of type, application, and region. On the basis of type, the market is classified into diagnostics, therapeutics, and software. The diagnostic segment is further categorized into magnetic resonance imaging, carotid ultrasound, cerebral angiography, electrocardiography, echocardiography, and others. The therapeutics segment is further divided into tissue plasminogen activator, anticoagulant, antiplatelet, and antihypertensive. As per application, the market is bifurcated into ischemic stroke and hemorrhagic stroke. Region-wise, the market is analyzed across North America (the U.S., Canada, and Mexico), Europe (Germany, France, the UK, Italy, Spain, and Rest of Europe), Asia-Pacific (China, Japan, Australia, India, South Korea, and Rest of Asia-Pacific), and LAMEA (Brazil, South Africa, Saudi Arabia, and Rest of LAMEA).
By Type
The stroke management market is segmented into diagnostics, therapeutics, and software. The diagnostic segment dominated the global market in 2022 and is expected to remain dominant during the Stroke Management Market Forecast period. This is attributed to increase in the diagnosis of stroke due to increase in awareness about early disease diagnosis of stroke. Further, surge in incidence of stroke has increased the demand for MRI, blood tests, CT angiography, which in turn drives the market growth. On the other hand, the software segment is expected to register the fastest CAGR growth during the forecast period, owing to development of advanced software for stroke management, and approval of new artificial intelligence integrated stroke solutions.
By Application
The stroke management market is bifurcated into ischemic stroke and hemorrhagic stroke. The ischemic stroke segment dominated the global Stroke Management Market Share in 2022 and is anticipated to continue this trend during the forecast period, owing to high prevalence of ischemic stroke. For instance, according to World Stroke Organization (WSO) 2022, more than 62% of all incident strokes are ischemic strokes. As per the same source, 7.6 million new ischemic stroke cases are diagnosed every year. However, hemorrhagic stroke is expected to be the fastest growing segment during the forecast period, owing to rising prevalence of intracerebral hemorrhagic stroke. For instance, according to the World Stroke Organization (WSO) 2022, more than 3.4 million new intracerebral hemorrhagic incidences are diagnosed every year.
By Region
The stroke management market is analyzed across North America, Europe, Asia-Pacific, and LAMEA. North America accounted for a major Stroke Management Market Share in 2022 and is expected to maintain its dominance during the forecast period.
The presence of several major players, such as Abbott Laboratories, VasSol, Inc., Brainomix, Medtronic plc, and Cardinal Health Inc. and the presence of well-developed healthcare infrastructure in the region drives the growth of the market. In addition, adoption of technologically advanced diagnostics further boosts the market growth in this region.
Asia-Pacific is expected to grow at the highest rate during the forecast period. The market growth in this region is attributed to increase in awareness about stroke in the general population. In addition, rise in adoption of stroke management further propels the market growth. Asia-Pacific offers profitable opportunities for key players operating in the stroke management market, thereby registering the fastest growth rate during the forecast period, owing to the rise in disposable income as well as increase in healthcare expenditure.

Competition Analysis
Competitive analysis and profiles of the major players in the stroke management, include Abbott Laboratories, Boston Scientific Corporation, Bristol-Myers Squibb Company, Cardinal Health, General Electric Company, Medtronic, Siemens AG, Aidoc, Brainomix, VasSol Inc., Qure.ai, Viz.ai, Inc., METHINKS, B Braun SE, iSchemaView, Inc., NeuraSignal, Inc., Deepnoid Co., Ltd., Heuron Co., Ltd., JLK, Koninklijke Philips N.V., and Merck & Co. Inc. Major players have adopted product launch, collaboration, product approval, and acquisition as key developmental strategies to improve their product portfolio and gain strong foothold in the stroke management market.
Recent Product Launch in the Stroke Management Market
In June 2020, iSchemaView, Inc. launched its RapidAI Insight. This platform offers solutions for systems to standardize stroke care process.
Recent Collaboration in the Stroke Management Market
GE Healthcare, a global medical device company, collaborated with Circle Neurovascular Imaging (Circle NVI), a medical device company. Under this agreement, NVI will provide AI-based tools to expand GE Healthcare’s FastStroke processing platform.
Recent Product Approval in the Stroke Management Market
In April 2023, iSchemaView, Inc. an AI driven medical technology company, received the Food and Drug Administration (FDA) approval for its Rapid NCCT Stroke. It is the first approved medical device for the detection of suspected intracranial hemorrhage and others.
In July 2022, Siemens Healthineers received the Food and Drug Administration (FDA) clearance for its MAGNETOM Free.Star, a magnetic resonance (MR) scanner designed to help improve patient access to MRI.
In June 2022, Heuron Co., Ltd., a medical technology company, received a Class 3 medical device approval for its cASPECTS AI software from Food and Drug Safety to assist stroke diagnosis.
Recent Acquisition in the Stroke Management Market
In May 2023, Siemens Healthineers and CommonSpirit Health acquired Block Imaging, a medical device company. This acquisition will help to provide more sustainable options for imaging parts and services and support increasing demand from the U.S. hospitals, health systems, and other care sites.
Recent Partnership in the Stroke Management Market
In April 2023, Medtronic partnered with AI innovator Qure.ai to revolutionize stroke management. Under this partnership, Qure's AI-powered solutions were integrated into primary stroke centers.
Key Benefits For Stakeholders
- This report provides a quantitative analysis of the market segments, current trends, estimations, and dynamics of the stroke management market analysis from 2022 to 2032 to identify the prevailing Stroke Management Market Opportunity.
- The market research is offered along with information related to key drivers, restraints, and opportunities.
- Porter's five forces analysis highlights the potency of buyers and suppliers to enable stakeholders make profit-oriented business decisions and strengthen their supplier-buyer network.
- In-depth analysis of the stroke management market segmentation assists to determine the prevailing market opportunities.
- Major countries in each region are mapped according to their revenue contribution to the global market.
- Market player positioning facilitates benchmarking and provides a clear understanding of the present position of the market players.
- The report includes the analysis of the regional as well as global stroke management market trends, key players, market segments, application areas, and market growth strategies.
Stroke Management Market Report Highlights
| Aspects | Details |
| Market Size By 2032 | USD 74 billion |
| Growth Rate | CAGR of 7.4% |
| Forecast period | 2022 - 2032 |
| Report Pages | 230 |
| By Type |
|
| By Application |
|
| By Region |
|
| Key Market Players | Merck & Co. Inc, METHINKS, Bristol-Myers Squibb Company, NeuraSignal, Inc., Qure.ai, B. Braun SE, Medtronic, iSchemaView, Inc., Koninklijke Philips N.V., Aidoc, JLK, Boston Scientific Corporation, VasSol Inc., Abbott Laboratories, Cardinal Health, Brainomix, Siemens AG, Viz.ai, Inc., Deepnoid Co., Ltd., Heuron Co., Ltd., General Electric Company |
Analyst Review
According to the insights of CXOs, growth of the global stroke management market is attributed to factors such as rise in new product launches, technological advancements, product approvals, and surge in collaboration between market key players to expand their businesses in different regions. For instance, in July 2022, Siemens Healthineers, a global medical technology company received the Food and Drug Administration (FDA) clearance for its MAGNETOM Free.Star, a whole-body magnetic resonance (MR) scanner designed to help improve patient access to MRI.
Furthermore, in October 2021, GE Healthcare, a global medical device company collaborated with Circle Neurovascular Imaging (Circle NVI) a medical device company. Under this agreement, NVI will provide AI-based tools to expand GE Healthcare’s FastStroke processing platform. In addition, Brainomix, an AI-powered medical solution company, entered a partnership with Visonable to enhance the delivery of care for stroke patients. Thus, all aforementioned factors drive the growth of the stroke management market.
The total market value of the Stroke Management Market is $36,091.5 million in 2022.
The forecast period in the report is from 2023 to 2032.
North America is the largest regional market for Stroke Management
There are 10 Stroke Management manufacturing companies are profiled in the report.
The top companies that hold the market share in Abbott Laboratories, Boston Scientific Corporation, Bristol-Myers Squibb Company, Cardinal Health, General Electric Company, and Medtronic.
The base year for the report is 2022.
Yes, Stroke Management companies are profiled in the report
Yes, the competitive landscape included in the Stroke Management Market report.
Loading Table Of Content...
Loading Research Methodology...



